Chapter 17: Q72P (page 850)
Draw the products of the following reactions:
Learning Materials
Features
Discover
Chapter 17: Q72P (page 850)
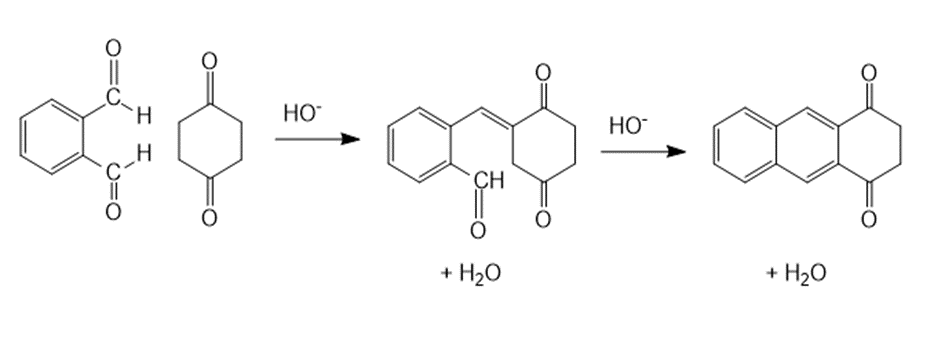
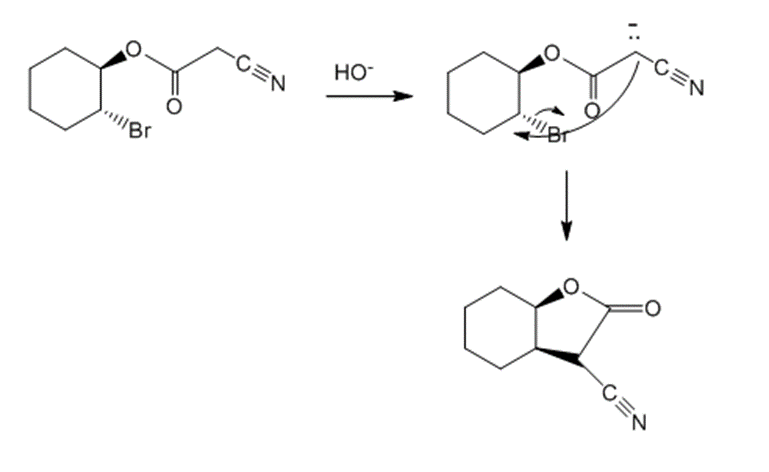
Draw the products of the following reactions:

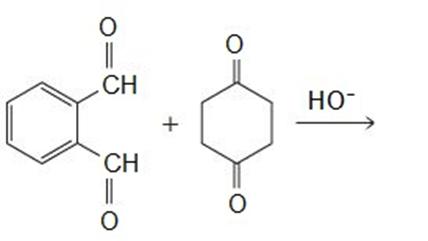
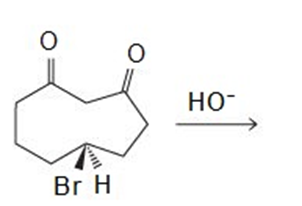
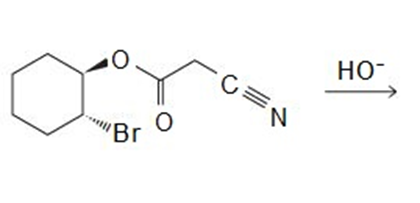
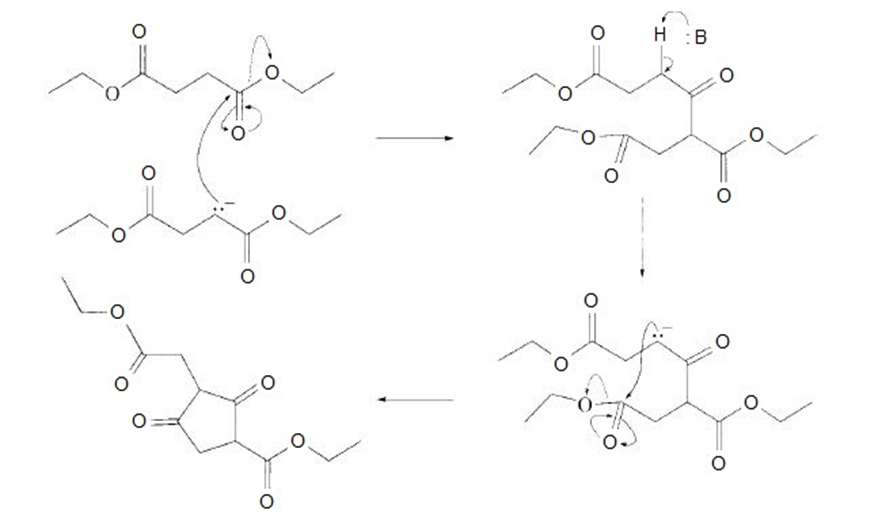

All the tools & learning materials you need for study success - in one app.
Get started for free
A racemic mixture of 2-methyl-1-phenyl-1-butanone is formed when (R)-2-methyl-1-phenyl-1-butanone is dissolved in an acidic or basic aqueous solution. Give an example of another ketone that undergoes acid- or base-catalyzed racemization.
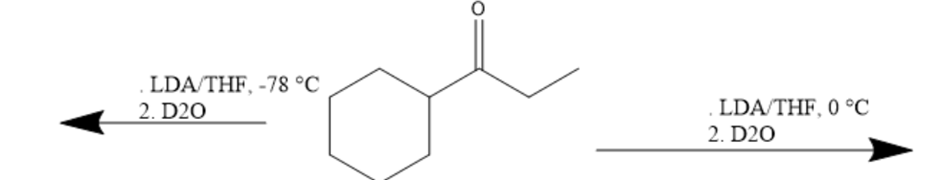
Draw the products of the following reactions:
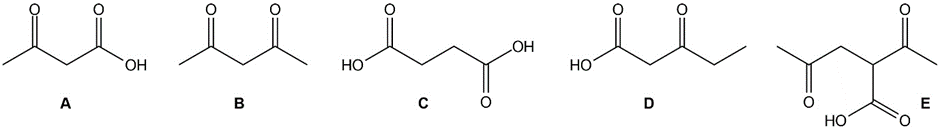
Which of the following compounds will decarboxylate when heated?
Using cyclopentanone as the reactant, show the product of a. acid-catalyzedketo–enolinterconversion. b. an aldol addition. c. an aldol condensation.
In the presence of excess base and excess halogen, a methyl ketone is converted to a carboxylate ion. The reaction is known as the haloform reaction because one of the products is haloform (chloroform, bromoform, or iodoform). Before spectroscopy became a routine analytical tool, the haloform reaction served as a test for methyl ketones: the formation of iodoform, a bright yellow compound, signaled that a methyl ketone was present. Why do only methyl ketones form a haloform?

What do you think about this solution?
We value your feedback to improve our textbook solutions.