Chapter 17: Q38P (page 833)
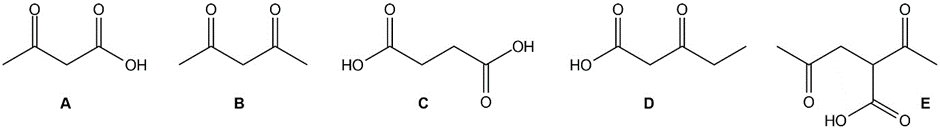
Which of the following compounds will decarboxylate when heated?
Short Answer
Compound showing decarboxylation is compound A and D, and E.
Learning Materials
Features
Discover
Chapter 17: Q38P (page 833)
Which of the following compounds will decarboxylate when heated?

Compound showing decarboxylation is compound A and D, and E.
All the tools & learning materials you need for study success - in one app.
Get started for free
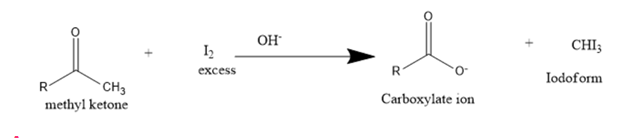
In the presence of excess base and excess halogen, a methyl ketone is converted to a carboxylate ion. The reaction is known as the haloform reaction because one of the products is haloform (chloroform, bromoform, or iodoform). Before spectroscopy became a routine analytical tool, the haloform reaction served as a test for methyl ketones: the formation of iodoform, a bright yellow compound, signaled that a methyl ketone was present. Why do only methyl ketones form a haloform?
Draw the products of the following reactions:
a. diethyl heptanedioate: (1) sodium ethoxide; (2) HCl
b. pentanoic acid + PBr3 + Br2, followed by hydrolysis
c. acetone + LDA/THF: (1) slow addition of ethyl acetate; (2) HCl
d. diethyl 2-ethylhexanedioate: (1) sodium ethoxide; (2) HCl
e. diethyl malonate: (1) sodium ethoxide; (2) isobutyl bromide; (3) HCl, H2O + ∆
f. acetophenone + LDA/THF: (1) slow addition of diethyl carbonate; (2) HCl
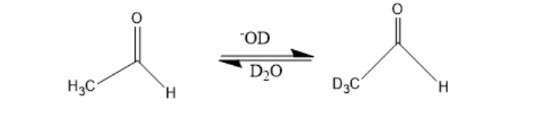
When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl hydrogens are exchanged with deuterium but the hydrogen attached to the carbonyl carbon is not.
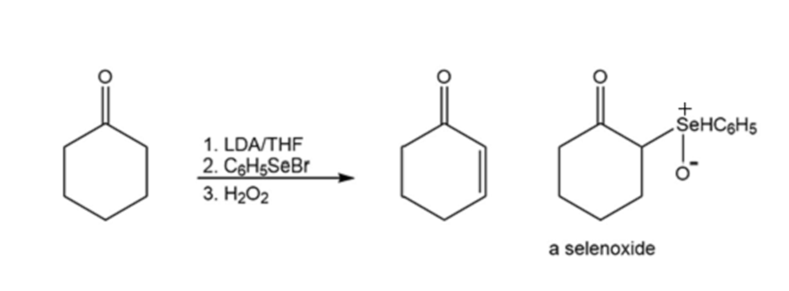
An -unsaturated carbonyl compound can be prepared by a reaction known as a selenenylation–oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction.
Can 2,4-pentanedione undergo an intramolecular aldol addition? If so, why? If not, why not?
What do you think about this solution?
We value your feedback to improve our textbook solutions.