Chapter 17: Q5P (page 805)
Rank the compounds in each of the following groups from strongest acid to weakest acid
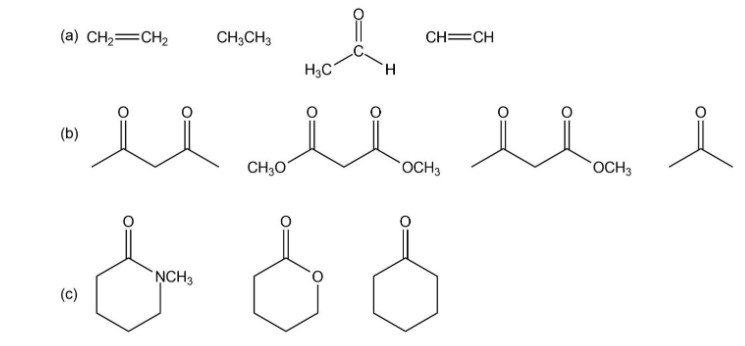
Short Answer
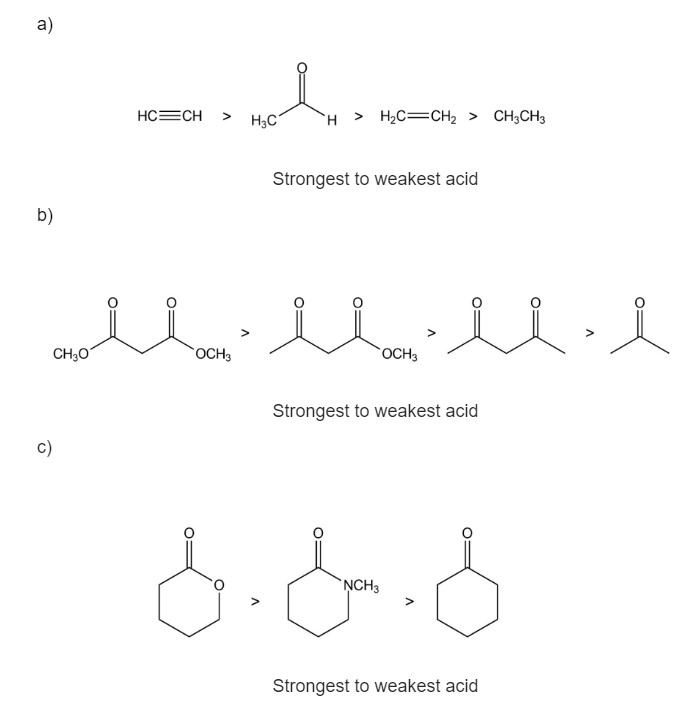
Learning Materials
Features
Discover
Chapter 17: Q5P (page 805)
Rank the compounds in each of the following groups from strongest acid to weakest acid


All the tools & learning materials you need for study success - in one app.
Get started for free
An -unsaturated carbonyl compound can be prepared by a reaction known as a selenenylation–oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction.
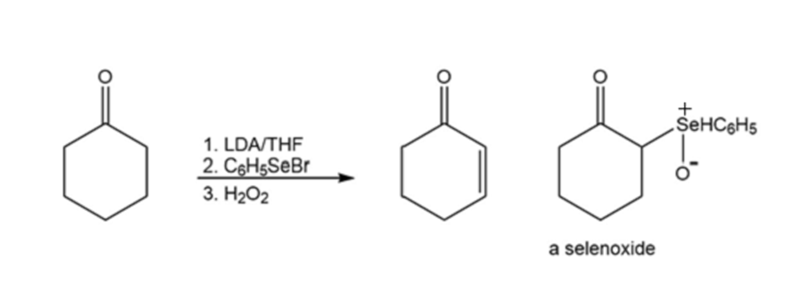
The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard reagent to add to the carbonyl group of an aldehyde or a ketone. Because the organozinc reagent is less reactive than a Grignard reagent, a nucleophilic addition to the ester group does not occur. The organozinc reagent is prepared by treating an ß-bromo ester with zinc.
Describe how each of the following compounds can be prepared, using a Reformatsky reaction:
What is the product of the following reaction?

Which of the following compounds decarboxylates when heated?
Starting with methyl propanoate, how could you prepare 4-methyl-3-heptanone?
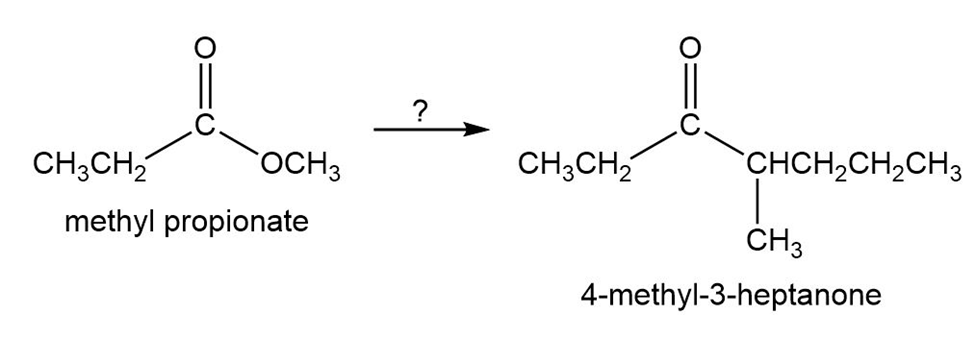
What do you think about this solution?
We value your feedback to improve our textbook solutions.