Chapter 17: Q32P (page 801)
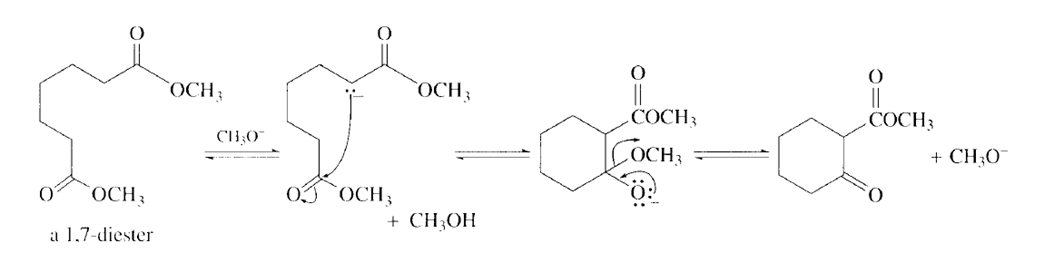
Write the mechanism for the reaction of a 1,7-diester with an alkoxide ion to form a cyclic -keto ester.
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q32P (page 801)
Write the mechanism for the reaction of a 1,7-diester with an alkoxide ion to form a cyclic -keto ester.

All the tools & learning materials you need for study success - in one app.
Get started for free
A ketone undergoes acid-catalyzed bromination, acid-catalyzed chlorination, racemization, and acid-catalyzed deuterium exchange at the -carbon. All of these reactions have similar rate constants. What does this tell you about the mechanisms of these reactions?
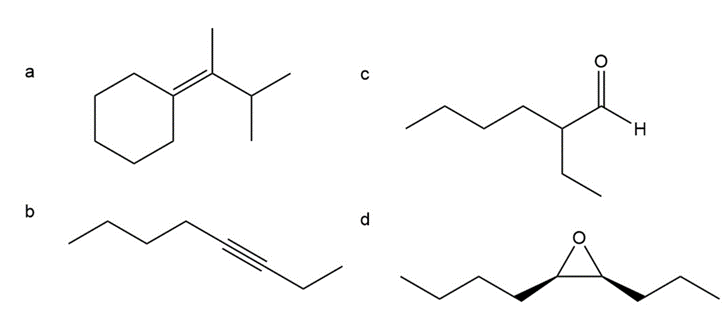
Describe how the following compounds can be prepared from compounds containing no more than six carbons. (You can also use triphenylphosphine.)
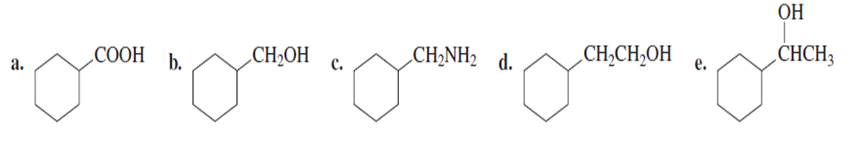
Problem: starting with bromocyclohexane, how can each of the following compounds be prepared?
How can you synthesize the following compounds from starting materials containing no more than four carbons?
(a)
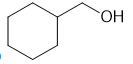
(b)
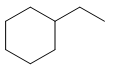
(c)
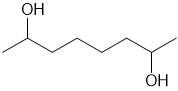
(d)
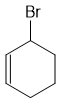
Which of the following compounds decarboxylates when heated?
What do you think about this solution?
We value your feedback to improve our textbook solutions.