Chapter 14: Q5P (page 626)
How could you distinguish the 1 H NMR spectra of the following compounds?

Short Answer
A)
 Structure with chemical shift values
Structure with chemical shift values
B)
 Structure with chemical shift values
Structure with chemical shift values
C)
 Structure with chemical shift values
Structure with chemical shift values
Learning Materials
Features
Discover
Chapter 14: Q5P (page 626)
How could you distinguish the 1 H NMR spectra of the following compounds?

A)
 Structure with chemical shift values
Structure with chemical shift values
B)
 Structure with chemical shift values
Structure with chemical shift values
C)
 Structure with chemical shift values
Structure with chemical shift values
All the tools & learning materials you need for study success - in one app.
Get started for free
How canNMR be used to prove that the addition of HBr to propene follows the rule that says that the electrophile adds to the sp2 carbon bonded to the most hydrogens?
Identify the compound with molecular formulaC3H7NO responsible for the 1H NMR spectrum shown here.

Determine the ratios of the chemically non-equivalent protons in a compound if the steps of the integration curves measure and , from left to right across the spectrum. Draw the structure of a compound whose NMR spectrum would show these integrals in the observed order.
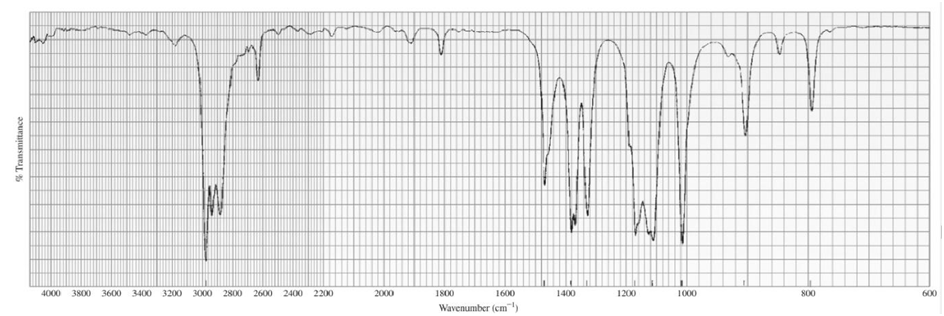
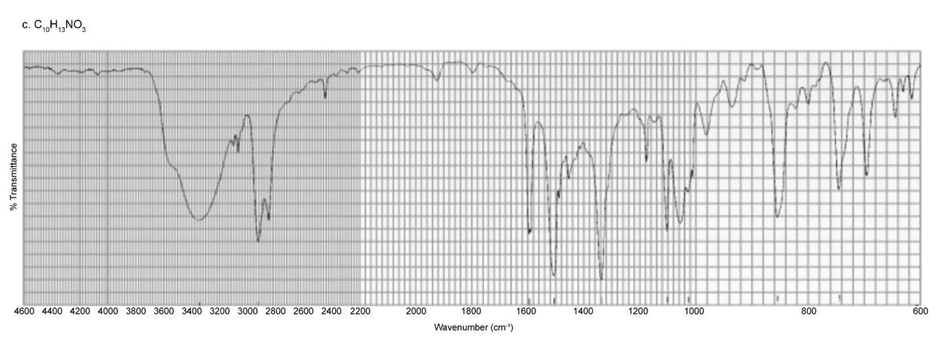
Identify each of the following compounds from its molecular formula and its IR and 1H NMR spectra:
a. C6H120
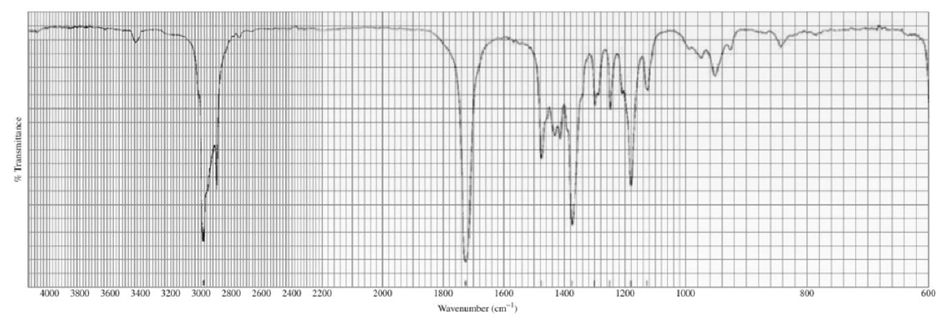
b.C6H14O
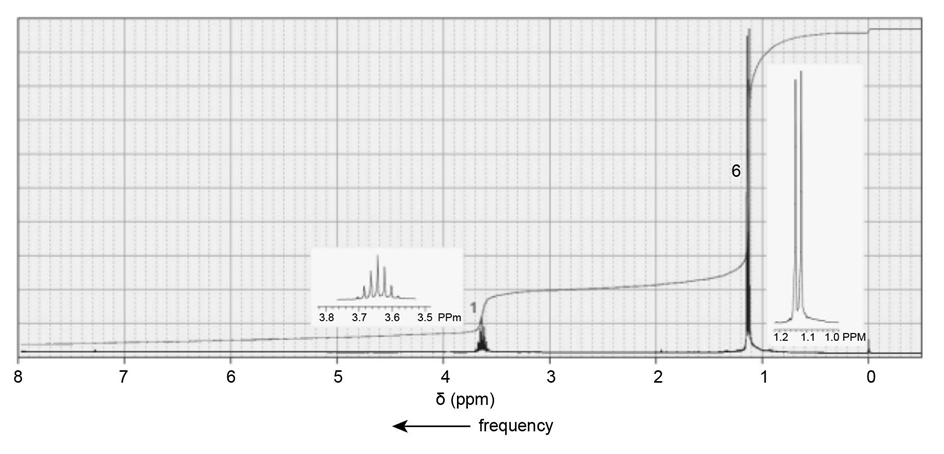
c. C10H13NO3

d. C11H14O2


Dr. N. M. Arr was called in to help analyze the 1 H NMR spectrum of a mixture of compounds known to contain only C, H, and Br. The mixture showed two singlets-one at 1.8 ppm and the other at 2.7 ppm-with relative integrals of 1: 6, respectively. Dr. Arr determined that the spectrum was that of a mixture of bromomethane and 2-Bromo-2-methylpropane. What was the ratio of bromomethane to 2-Bromo-2-methylpropane in the mixture?
What do you think about this solution?
We value your feedback to improve our textbook solutions.