Chapter 14: Q14-73P (page 620)
Calculate the amount of energy (in calories) required to flip an 1Hnucleus in an NMR spectrometer that operates at 300 MHz.
Short Answer
The amount of energy required to flip1H nucleus is 475.16 X 10-28
Learning Materials
Features
Discover
Chapter 14: Q14-73P (page 620)
Calculate the amount of energy (in calories) required to flip an 1Hnucleus in an NMR spectrometer that operates at 300 MHz.
The amount of energy required to flip1H nucleus is 475.16 X 10-28
All the tools & learning materials you need for study success - in one app.
Get started for free
How many signals are produced by each of the following compounds in its
a.
b. NMR spectrum?
1.
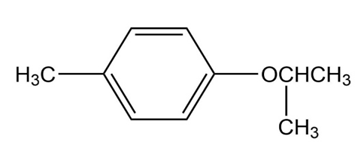
2.
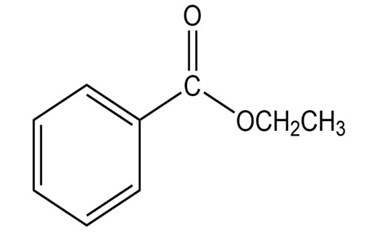
3.
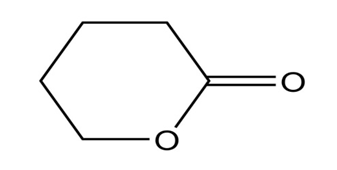
4.
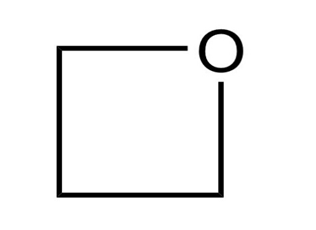
5.
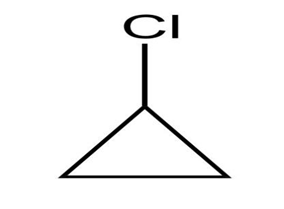
6.

Determine the ratios of the chemically non-equivalent protons in a compound if the steps of the integration curves measure and , from left to right across the spectrum. Draw the structure of a compound whose NMR spectrum would show these integrals in the observed order.
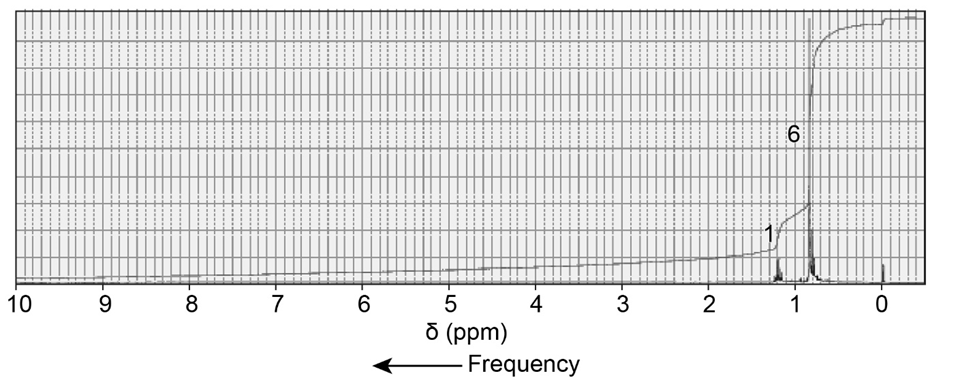
Identify the compound with molecular formula C6H14 that is responsible for the following 1H NMR spectrum:
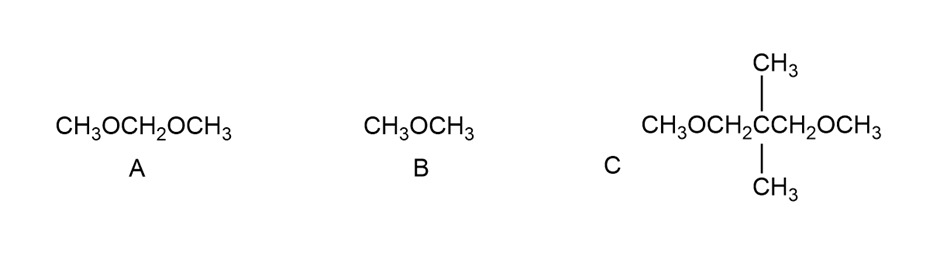
How could you distinguish the 1 H NMR spectra of the following compounds?
The followingNMR spectra are for four compounds, each with a molecular formula of . Identify the compounds.
a.
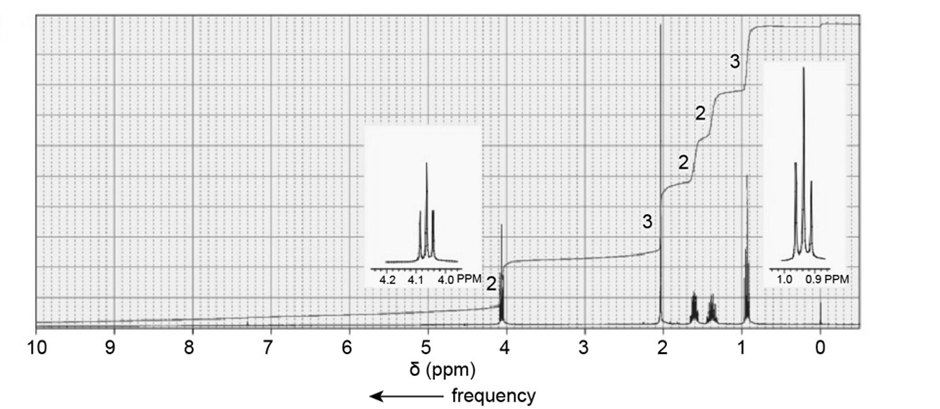
b.
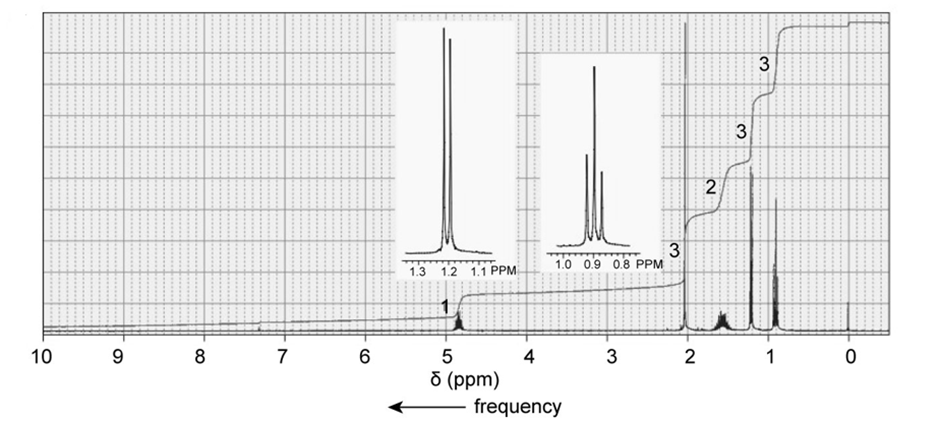
c.
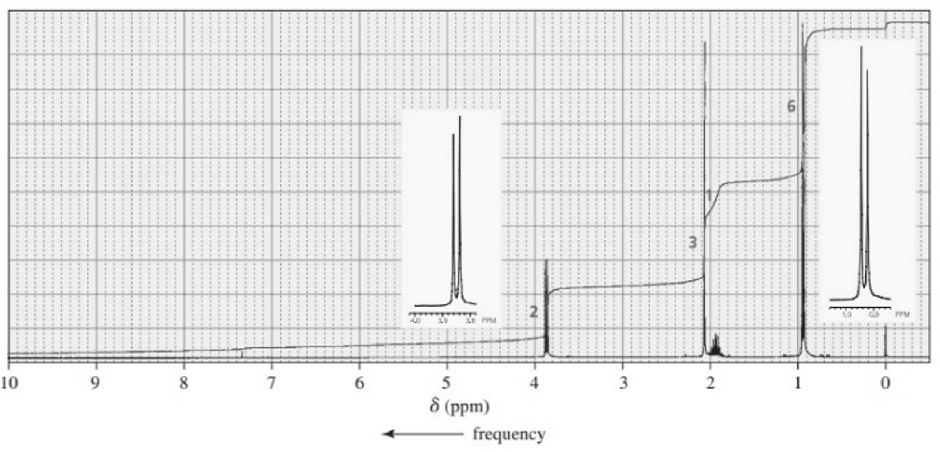
d.

What do you think about this solution?
We value your feedback to improve our textbook solutions.