Chapter 19: Q28P (page 946)
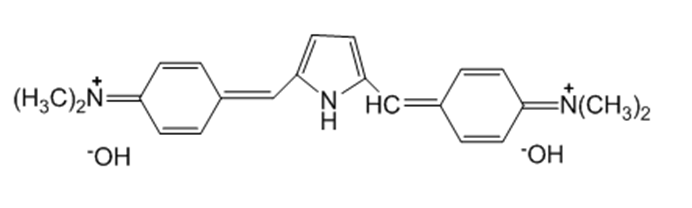
Pyrrole reacts with excess para-(N,N-dimethylamino)benzaldehyde to form a highly colored compound. Draw the structure of the colored compound.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q28P (page 946)
Pyrrole reacts with excess para-(N,N-dimethylamino)benzaldehyde to form a highly colored compound. Draw the structure of the colored compound.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:
a.Propose a mechanism for the following reaction:
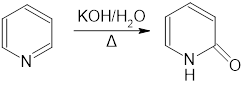
b.What other product is formed in this reaction?
Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a Lewis acid such as .
a. What are the major and minor products of the following reaction?
b. Would you expect epoxides to undergo similar reactions?

Rank the following compounds from strongest acid to weakest acid:

Propose a mechanism for the following reactions:
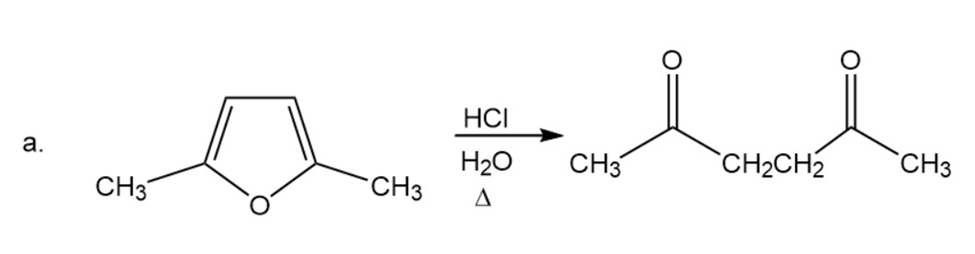
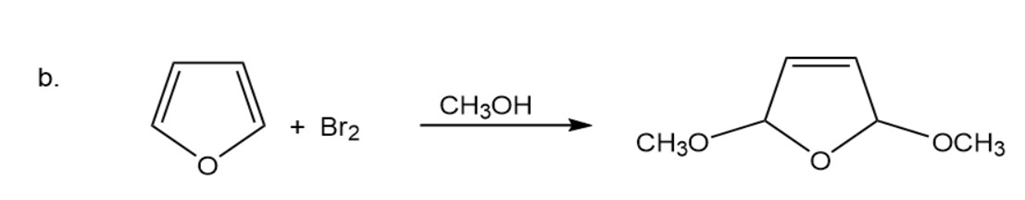
The chemical shifts of the C-2 hydrogen in the spectra of pyrrole, pyridine, and pyrrolidine are 2.82 ppm, 6.42 ppm, and 8.50 ppm. Match each heterocycle with its chemical shift.
What do you think about this solution?
We value your feedback to improve our textbook solutions.