Chapter 19: Q23P (page 946)
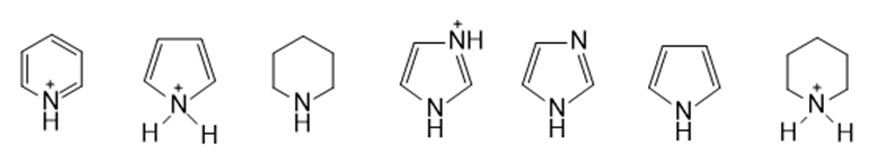
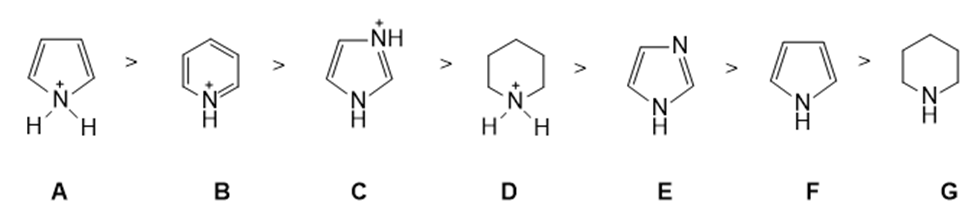
Rank the following compounds from strongest acid to weakest acid:
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q23P (page 946)
Rank the following compounds from strongest acid to weakest acid:


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Name the following:
a.
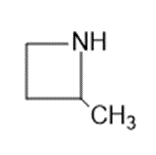
b.
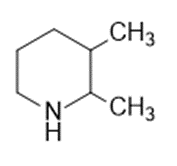
c.
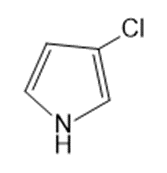
d.
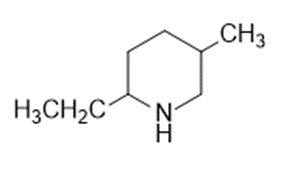
Question:
a.Propose a mechanism for the following reaction:
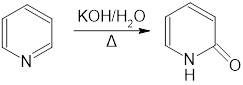
b.What other product is formed in this reaction?
Quinolines, heterocyclic compounds that contain a pyridine ring fused to a benzene ring, are commonly synthesized by a method known as the Skraup synthesis, in which aniline reacts with glycerol under acidic conditions. Nitrobenzene is added to the reaction mixture to serve as an oxidizing agent.
The first step in the synthesis is the dehydration of glycerol to propenal.
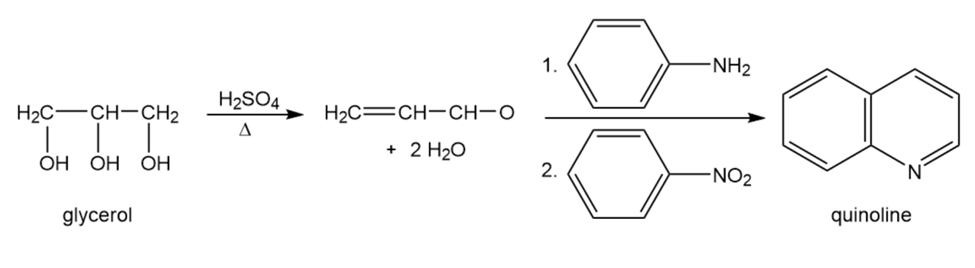
a. What product would be obtained if para-ethylaniline were used instead of aniline?
b. What product would be obtained if 3-hexen-2-one were used instead of glycerol?
c. What starting materials are needed for the synthesis of 2,7-diethyl-3-methylquinoline?
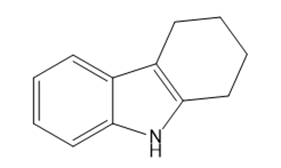
What starting materials are required to synthesize the following compounds, using the Fischer indole synthesis?
a.
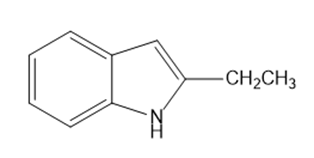
b.
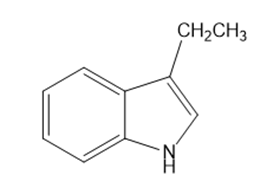
c.
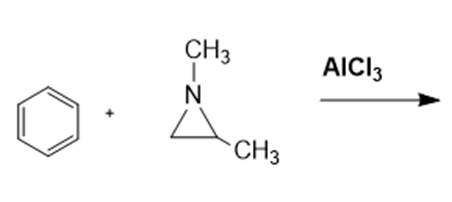
Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a Lewis acid such as .
a. What are the major and minor products of the following reaction?
b. Would you expect epoxides to undergo similar reactions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.