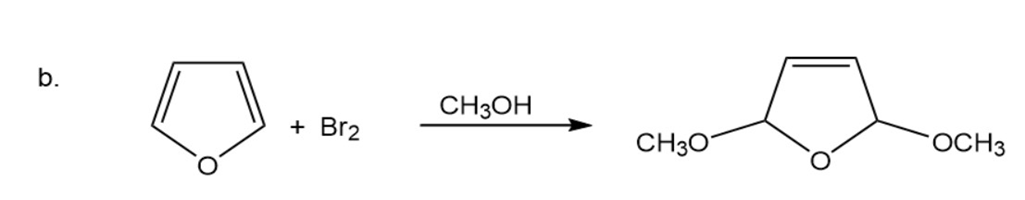
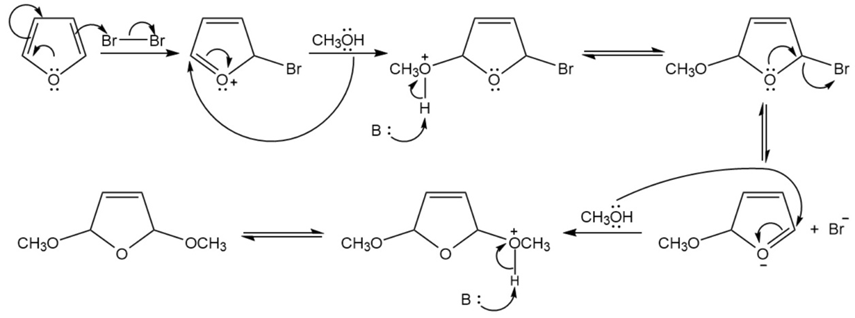
Chapter 19: Q39P (page 948)
Propose a mechanism for the following reactions:

Short Answer
(a)
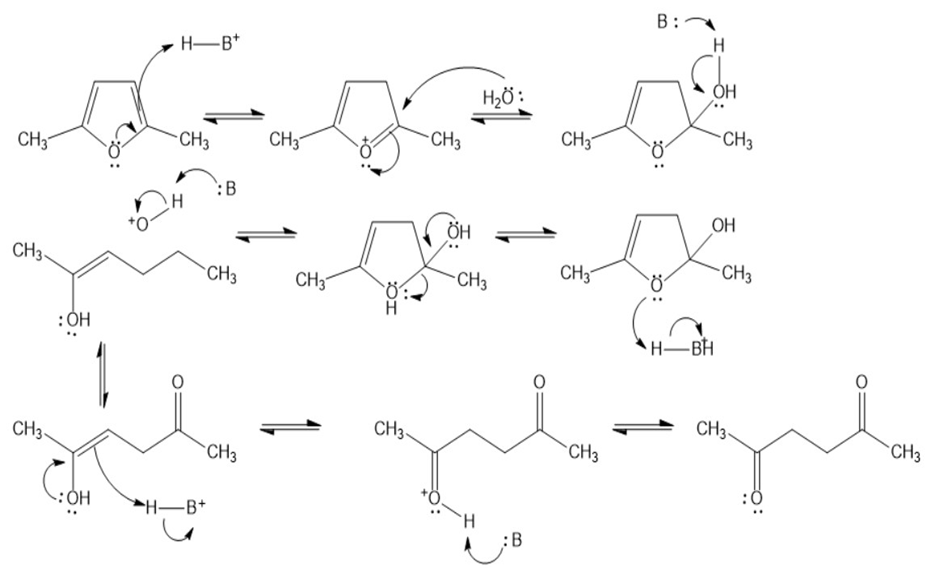
(b)

Learning Materials
Features
Discover
Chapter 19: Q39P (page 948)
Propose a mechanism for the following reactions:


(a)

(b)


All the tools & learning materials you need for study success - in one app.
Get started for free
Why is the conjugate acid of morpholine more acidic than the conjugate acid of piperidine?
Why is protonated pyrimidine more acidic than protonated pyridine ?
Question:
a.Propose a mechanism for the following reaction:
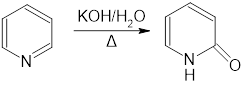
b.What other product is formed in this reaction?
Rank imidazole, pyrrole, and benzene from most reactive to least reactive toward electrophilic aromatic substitution.
Organic chemists work with tetraphenylporphyrins rather than with porphyrins because tetraphenylporphyrins are much more resistant to air oxidation. Tetraphenylporphyrin can be prepared by the reaction of benzaldehyde with pyrrole. Propose a mechanism for the formation of the ring system shown here:

What do you think about this solution?
We value your feedback to improve our textbook solutions.