Chapter 8: Q 72P (page 376)
Classify the following species as aromatic, nonaromatic, or antiaromatic:
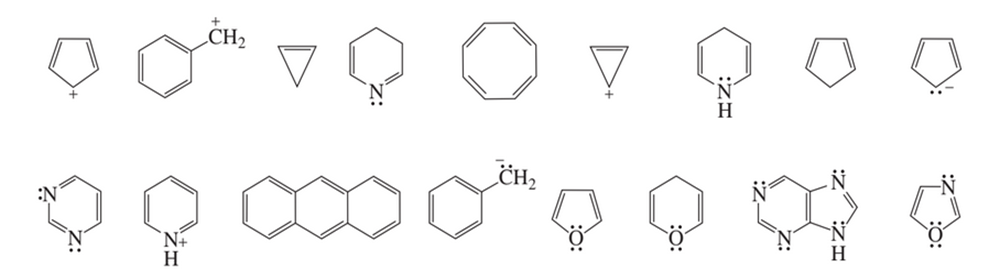
Short Answer
Aromatic:

Antiaromatic:

Neither aromatic nor antiaromatic:
Learning Materials
Features
Discover
Chapter 8: Q 72P (page 376)
Classify the following species as aromatic, nonaromatic, or antiaromatic:

Aromatic:

Antiaromatic:

Neither aromatic nor antiaromatic:

All the tools & learning materials you need for study success - in one app.
Get started for free
How would the following substituents affect the rate of a Diels–Alder reaction?
a. an electron-donating substituent in the diene
b. an electron-donating substituent in the dienophile
c. an electron-withdrawing substituent in the diene
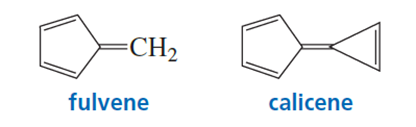
a. In what direction is the dipole moment in fulvene? Explain.
b. In what direction is the dipole moment in calicene? Explain.
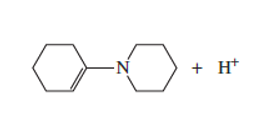
Which atom in the following compound is most likely to be protonated?
Draw the product or products that would be obtained from each of the following reactions:

Which of the following is the strongest acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.