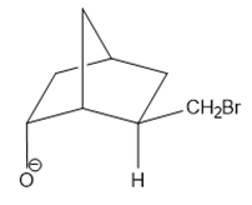
Chapter 22: Q29P (page 1060)
Which of the following two compounds eliminates HBr more rapidly in a basic solution?

Short Answer
The answer is,
Learning Materials
Features
Discover
Chapter 22: Q29P (page 1060)
Which of the following two compounds eliminates HBr more rapidly in a basic solution?

The answer is,

All the tools & learning materials you need for study success - in one app.
Get started for free
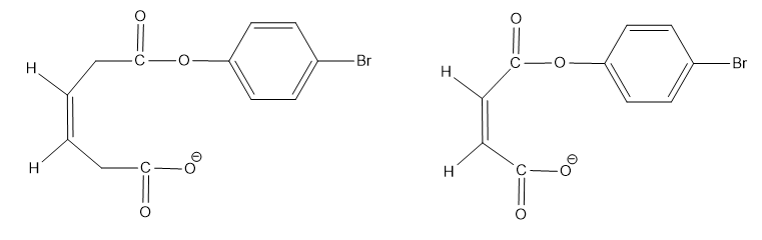
Which compound forms an anhydride more rapidly?
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6M-1S-1 . in the presence of \(C{o^{2 + }}\), the rate constant is \(1.5 \times 1{0^{\bf{6}}}{M^{ - 1}}{s^{ - 1}}\). What rate enhancement does the catalyst provide?
The deuterium kinetic isotope effect for the hydrolysis of aspirin is 2.2. What does this tell you about the kind of catalysis exerted by the o- carboxyl substituent?
Aldolate shows no activity if it is incubated with iodoacetic acid before fructose-1,6-bisphosphate is added to the reaction mixture. What causes this loss of activity?
Which of the following amino acid side chains can form an imine with a substrate?

What do you think about this solution?
We value your feedback to improve our textbook solutions.