Chapter 22: Q26P (page 1059)
Which of the following amino acid side chains can form an imine with a substrate?

Short Answer
The answer is
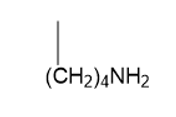
Learning Materials
Features
Discover
Chapter 22: Q26P (page 1059)
Which of the following amino acid side chains can form an imine with a substrate?

The answer is
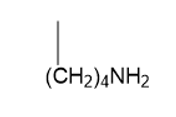
All the tools & learning materials you need for study success - in one app.
Get started for free
Which compound has the greatest rate of hydrolysis at pH=3.5:benzamide, o-carboxybenzamide, o-formylbenzamide, or o-hydroxybenzamide?
In glycolysis, why must glucose-6-phosphate isomerizes to fructose- 6- phosphate before the cleavage reaction with aldose occurs?
a. Explain why the alkyl halide shown here reacts much more rapidly with guanine than does a primary alkyl halide (such as pentyl chloride).

b. The alkyl halide can react with two guanines, each in a different DNA chain, thereby cross-linking the chains. Propose a mechanism for the cross-linking reaction.
Which of the following two compounds eliminates HBr more rapidly in a basic solution?

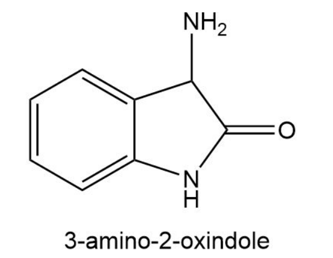
3-Amino-2-oxindole catalyzes the decarboxylation of -keto acids.
a. Propose a mechanism for the catalyzed reaction.
b. Would 3-aminoindole be equally effective as a catalyst?
What do you think about this solution?
We value your feedback to improve our textbook solutions.