Chapter 22: Q33P (page 1060)
The deuterium kinetic isotope effect for the hydrolysis of aspirin is 2.2. What does this tell you about the kind of catalysis exerted by the o- carboxyl substituent?
Short Answer
The answer is,
General- base catalyst
Learning Materials
Features
Discover
Chapter 22: Q33P (page 1060)
The deuterium kinetic isotope effect for the hydrolysis of aspirin is 2.2. What does this tell you about the kind of catalysis exerted by the o- carboxyl substituent?
The answer is,
General- base catalyst
All the tools & learning materials you need for study success - in one app.
Get started for free
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6M-1S-1 . in the presence of \(C{o^{2 + }}\), the rate constant is \(1.5 \times 1{0^{\bf{6}}}{M^{ - 1}}{s^{ - 1}}\). What rate enhancement does the catalyst provide?
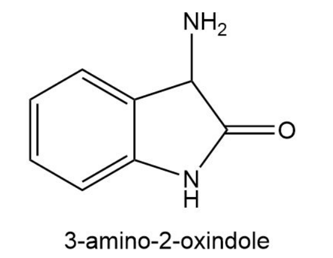
3-Amino-2-oxindole catalyzes the decarboxylation of -keto acids.
a. Propose a mechanism for the catalyzed reaction.
b. Would 3-aminoindole be equally effective as a catalyst?
Which compound has the greatest rate of hydrolysis at pH=3.5:benzamide, o-carboxybenzamide, o-formylbenzamide, or o-hydroxybenzamide?
Which of the following amino acid side chains can form an imine with a substrate?
In glycolysis, why must glucose-6-phosphate isomerizes to fructose- 6- phosphate before the cleavage reaction with aldose occurs?
What do you think about this solution?
We value your feedback to improve our textbook solutions.