Chapter 26: Q11P (page 1332)
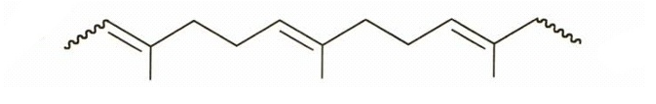
. (a) Draw the structure of gutta-percha, a natural rubber with all its double bonds in the trans configuration.
(b) Suggest why gutta-percha is not very elastic, even after it is vulcanized.
Short Answer
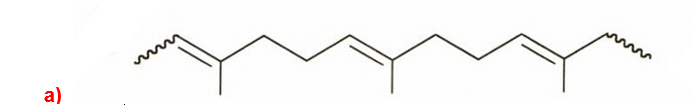
b)Due to more trans double bonds in gutta-percha, there occurs higher degree of crystallinity and lesser elasticity.