Chapter 26: Q25P (page 1342)
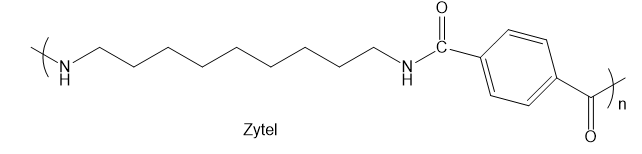
Zytel®finds extensive use in the automotive and electronics industries. Zytel® has the structure shown.
- What functional group is contained in the Zytel® structure?
- Is Zytel® a chain-growth polymer or a step-growth polymer?
- Draw the products that would be formed if the polymer were completely hydrolyzed under acidic or basic conditions.
Short Answer
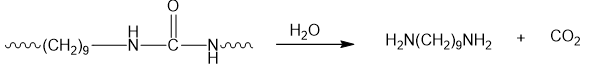
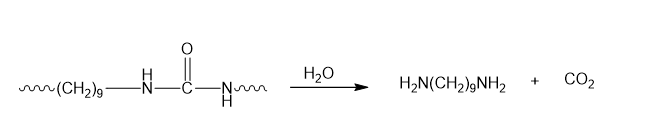
- Zytel is a polyurea.
- A polyurea is a step-growth polymer.