Chapter 26: Q5P (page 1328)

Short Answer
(a) Vinyl chloride polymerizes well.
(b) Vinyl acetate does not polymerize well.
(c) does not possible polymerize well.
Learning Materials
Features
Discover
Chapter 26: Q5P (page 1328)

(a) Vinyl chloride polymerizes well.
(b) Vinyl acetate does not polymerize well.
(c) does not possible polymerize well.
All the tools & learning materials you need for study success - in one app.
Get started for free
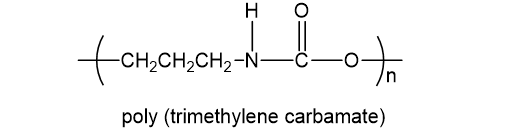
. Poly (trimethylene carbamate) is used in high-quality synthetic leather. It has the structure shown.
(a) What type of polymer is poly (trimethylene carbamate)?
(b) Is this a chain-growth polymer or a step-growth polymer?
(c) Draw the products that would be formed if the polymer were completely hydrolyzed under acidic or basic conditions.
(a) Isobutylene and isoprene coplymerize to give “butyl rubber”. Draw the structure of the repeating unit in butyl rubber, assuming that the two monomers alternate.
(b) Styrene and butadiene copolymerize to form styrene-butadiene rubber (SBR) for automobile tires. Draw the structure of the repeating unit in SBR, assuming that the two monomers alternate.
. (a) Draw the structure of gutta-percha, a natural rubber with all its double bonds in the trans configuration.
(b) Suggest why gutta-percha is not very elastic, even after it is vulcanized.
Explain why the addition of a small amount of glycerol to the polymerization mixture gives a stiffer urethane foam.
Poly (vinyl alcohol), a hydrophilic polymer used in aqueous adhesives, is made by polymerizing vinyl acetate and then hydrolyzing the ester linkages.
(a) Give the structures of poly (vinyl acetate) and poly (vinyl alcohol).
(b) Vinyl acetate is an ester. Is poly (vinyl acetate) therefore a polyester? Explain.
(c) We have seen that basic hydrolysis destroys the Dacron polymer. Poly (vinyl acetate) is converted to poly (vinyl alcohol) by a basic hydrolysis of the ester groups. Why doesn’t the hydrolysis destroy the poly (vinyl alcohol) polymer?
(d) Why is poly (vinyl alcohol) made by this circuitous route? Why not just polymerize vinyl alcohol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.