Chapter 26: Q10P (page 1330)
Draw the structure of isotactic poly(acrylonitrile) and syndiotactic polystyrene.
Short Answer
1)
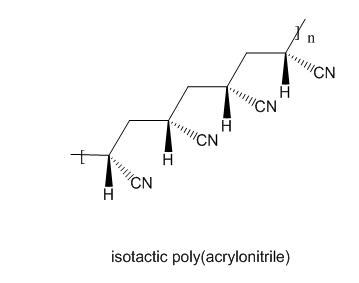
2)
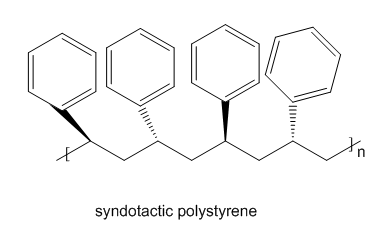
Learning Materials
Features
Discover
Chapter 26: Q10P (page 1330)
Draw the structure of isotactic poly(acrylonitrile) and syndiotactic polystyrene.
1)

2)

All the tools & learning materials you need for study success - in one app.
Get started for free
Chain branching is not as common with anionic polymerization as it is with free-radical polymerization and cationic polymerization.
Bisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction. (Hint: This is a condensation because three molecules are joined with loss of water. The mechanism belongs to another class of reactions though.)
(a) Isobutylene and isoprene coplymerize to give “butyl rubber”. Draw the structure of the repeating unit in butyl rubber, assuming that the two monomers alternate.
(b) Styrene and butadiene copolymerize to form styrene-butadiene rubber (SBR) for automobile tires. Draw the structure of the repeating unit in SBR, assuming that the two monomers alternate.
(a) Nomex®, a strong fire-resistant fabric, is a polyamide made from meta-phthalic acid and meta-diaminobenzene. Draw the structure of Nomex.
(b) Kevlar®, made from terephthalic acid (para-phthalic acid) and para-diaminobenzene, is used in making tire cord and bulletproof vests. Draw the structure of Kevlar.

What do you think about this solution?
We value your feedback to improve our textbook solutions.