Chapter 26: Q3P (page 1326)
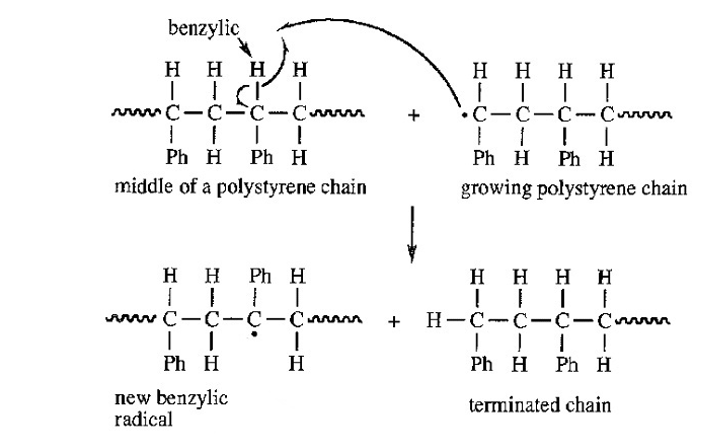
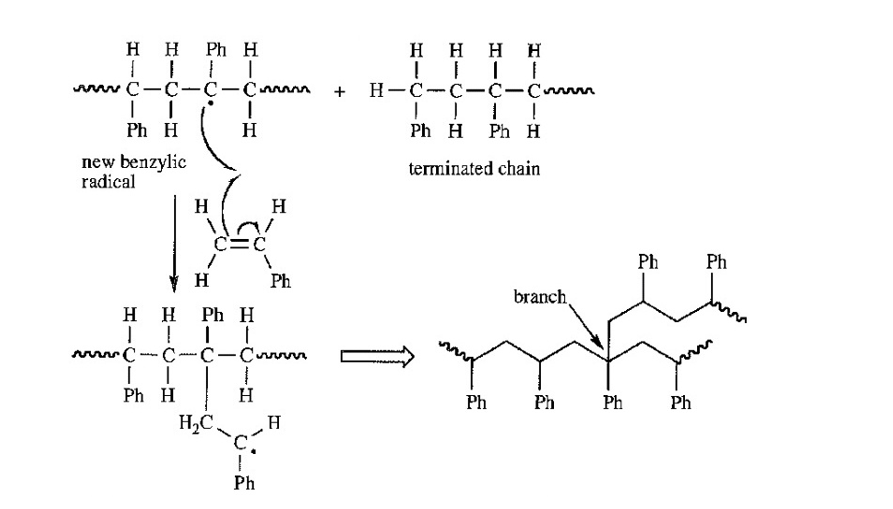
Give a mechanism, using Figure 26-1 as a guide, showing chain branching during the free-radical polymerization of styrene. There are two types of aliphatic hydrogens in the polystyrene chain. Which type is more likely to be abstracted?
Short Answer
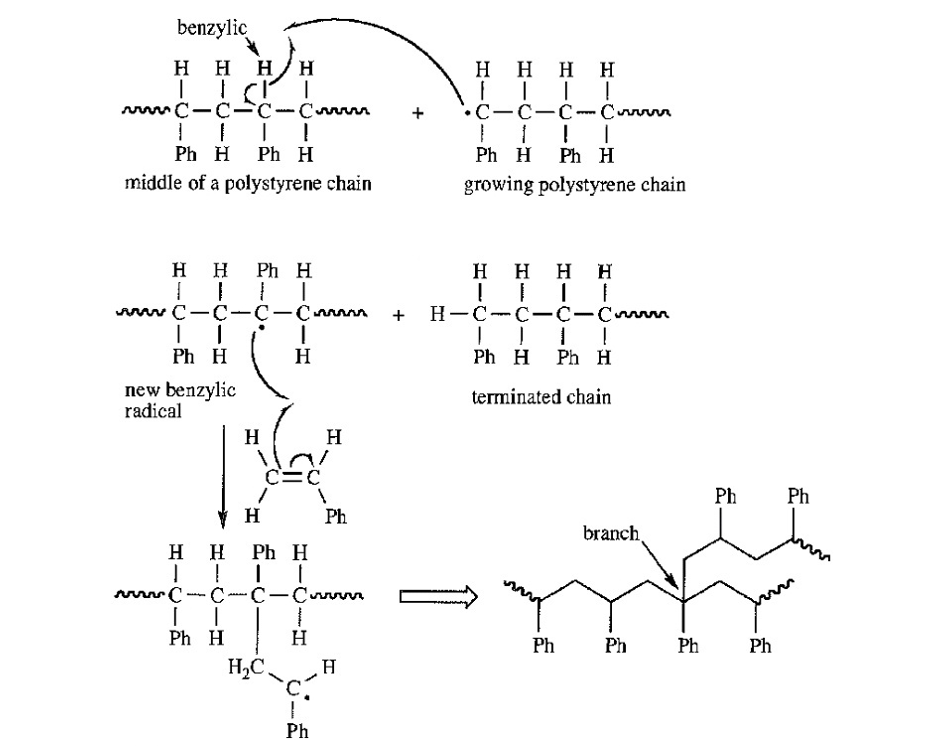
That hydrogen in the polystyrene chain will be abstracted after which radical becomes stable. Benzylic hydrogen will get abstracted and not secondary hydrogen as benzylic radical is tertiary as well as resonance stabilized.