Chapter 7: Q 28P (page 369)
Predict the elimination products of the following reactions and label the major products.
- Cis-1-bromo-2-methylcyclohexane +
in
- Trans-1-bromo-2-methylcyclohexane +
in
Short Answer
a.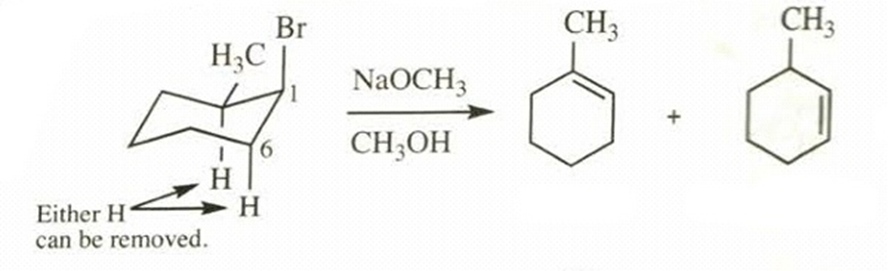
Major Minor
b.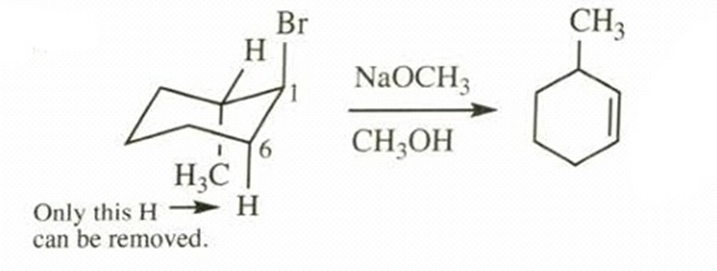
Major
Learning Materials
Features
Discover
Chapter 7: Q 28P (page 369)
Predict the elimination products of the following reactions and label the major products.
a.
Major Minor
b.
Major
All the tools & learning materials you need for study success - in one app.
Get started for free
Finish Solved Problem 7-3 by showing how the rearranged carbocations give the four products shown in the problem. Be careful when using curved arrows to show deprotonation and/or nucleophilic attack by the solvent. The curved arrows always show movement of electrons, not movement of protons or other species.
Give the substitution and elimination products you would expect from the following reactions.
Question: Draw five more compounds of formula.
Show what happens in step-2 of the example if the solvent acts as a nucleophile (forming a bond to carbon) rather than as a base (removing a proton).
(a) Design an alkyl halide that will give only 2,4-diphenylpent-2-ene upon treatment with potassium tert-butoxide (a bulky base that promotes E2 elimination).
(b) What stereochemistry is required in your alkyl halide so that only the following stereoisomer of the product is formed?
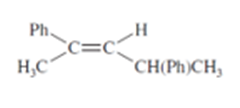
What do you think about this solution?
We value your feedback to improve our textbook solutions.