Chapter 7: Q 20P (page 360)
Give the substitution and elimination products you would expect from the following reactions.
- 3-bromo-3-ethylpentane heated in methanol
- 1-iodo-1-phenylcyclopentane heated in ethanol
- 1-bromo-2-methylcyclohexane + silver nitrate in water (
forces ionization)
Short Answer
a.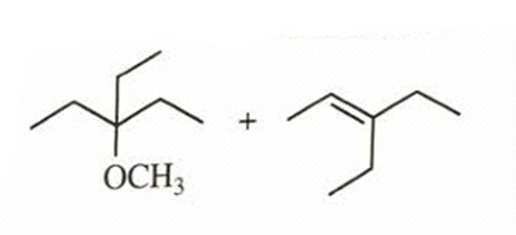
Substitution Elimination
Product Product
b.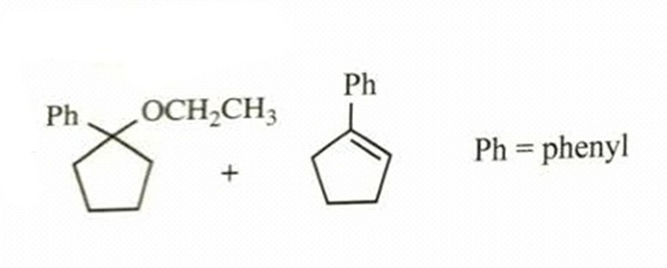
Substitution Elimination
Product Product
c.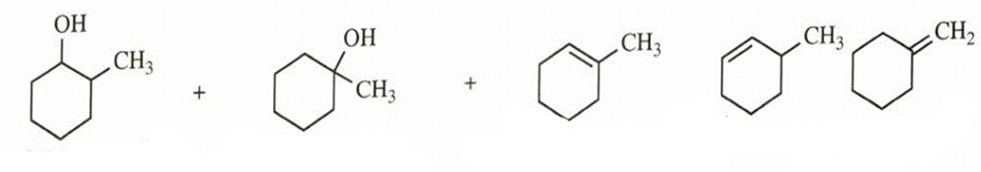

Elimination Minor
Product product product
With rearrangement
 reaction occurs as tertiary carbocation is formed when leaving group bromine leaves. Then, methanol acts as nucleophile and attacks carbocation which leads to formation of substitution product. Elimination product is also possible, because after carbocation formation, the adjacent carbon’s hydrogen can stabilize positive charge on carbocation by forming double bond. E1 product will be obtained.
reaction occurs as tertiary carbocation is formed when leaving group bromine leaves. Then, methanol acts as nucleophile and attacks carbocation which leads to formation of substitution product. Elimination product is also possible, because after carbocation formation, the adjacent carbon’s hydrogen can stabilize positive charge on carbocation by forming double bond. E1 product will be obtained.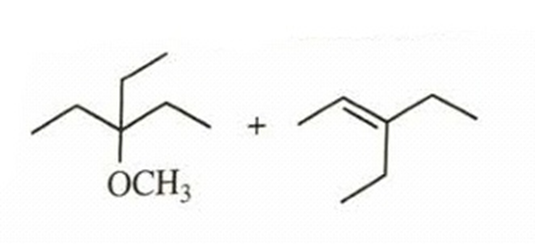
 reaction occurs as tertiary carbocation is formed when leaving group iodine leaves. Then, ethanol acts as nucleophile and attacks carbocation and this leads to formation of substitution product. Elimination product is also possible, because after carbocation formation, the adjacent carbon’s hydrogen can stabilize positive charge; and ethanol abstracts this proton and forms a double bond. E1 product will be obtained.
reaction occurs as tertiary carbocation is formed when leaving group iodine leaves. Then, ethanol acts as nucleophile and attacks carbocation and this leads to formation of substitution product. Elimination product is also possible, because after carbocation formation, the adjacent carbon’s hydrogen can stabilize positive charge; and ethanol abstracts this proton and forms a double bond. E1 product will be obtained.