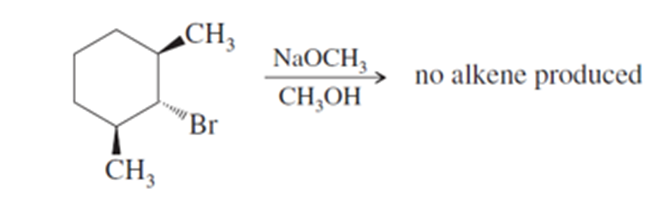
Chapter 7: Q 26P (page 367)
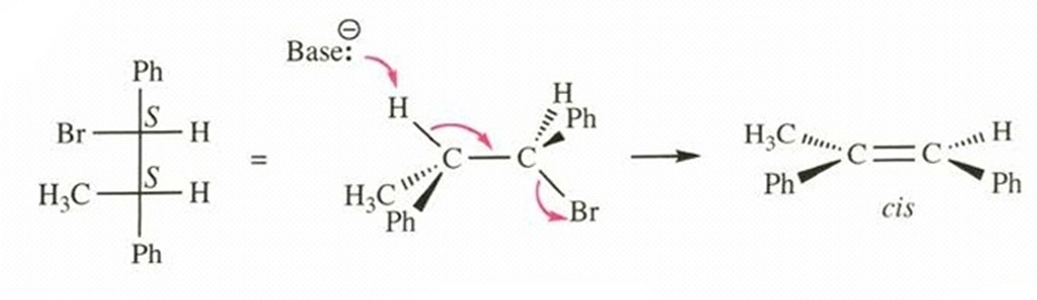
Show that the (S,S) enantiomer of this (R,R) diastereomer of 1-bromo-1,2-diphenylpropane also undergoes E2 elimination to give the cis diastereomer of the product. (We do not expect these achiral reagents to distinguish between enantiomers.)
Short Answer
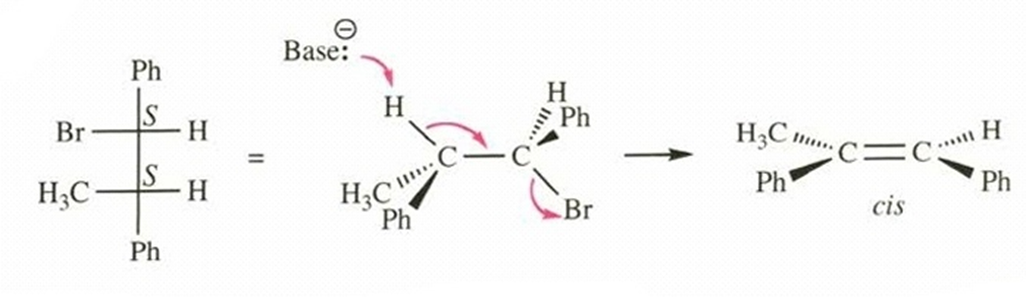
(S,S)-1-bromo-1,2-diphenylpropane