Chapter 7: Q61P (page 392)
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown.
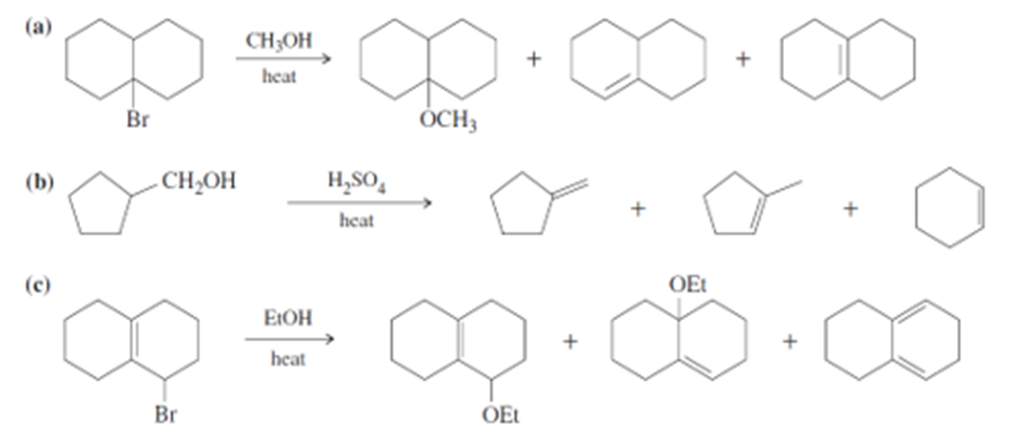
Short Answer
(a) 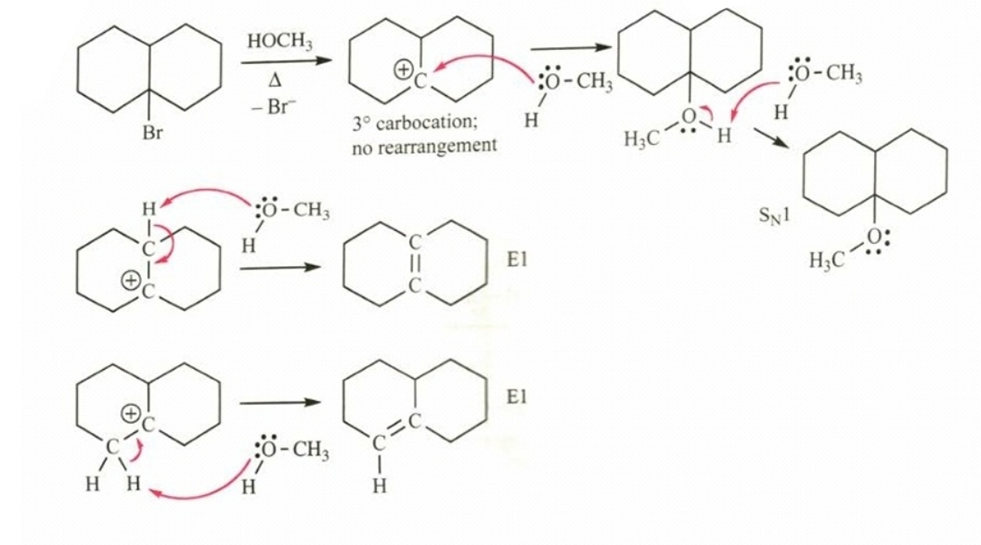
(b) 
(c) 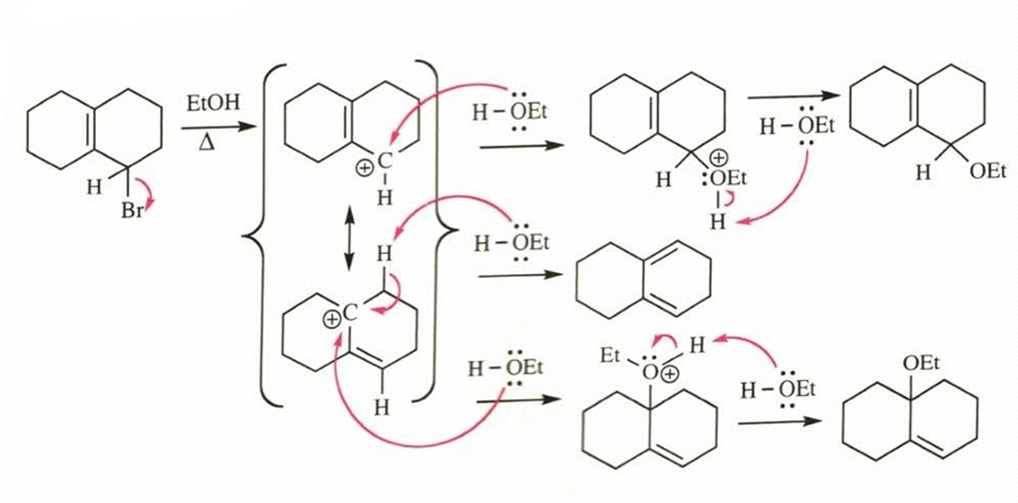
Learning Materials
Features
Discover
Chapter 7: Q61P (page 392)
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown.

(a) 
(b) 
(c) 
All the tools & learning materials you need for study success - in one app.
Get started for free
For each reaction, decide whether substitution or elimination (or both) is possible, and predict the product you expect. Label the major products.
(a) 1 - bromo - 1 - methylcyclohexane + NaOH in acetone
(b) 1 - bromo - 1 - methylcyclohexane + triethylamine (Et3N:)
(c) chlorocyclohexane + NaOCH3 in CH3OH
(d) chlorocyclohexane + NaOC(CH3) in (CH3)3COH
When 2-bromo-3-phenylbutane is treated with sodium methoxide, two alkenes result (by E2 elimination). The Zaitsev product predominates.
Question: Give systematic (IUPAC) names of the following alkenes.
(a)
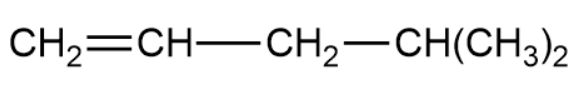
(b)
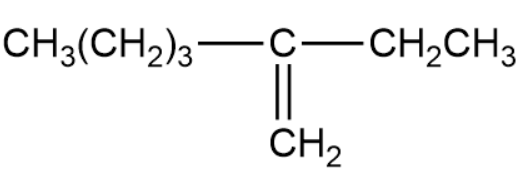
(c)

(d)
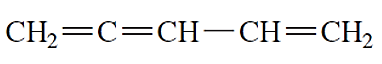
(e)
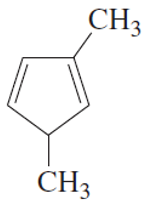
(f)

(g)
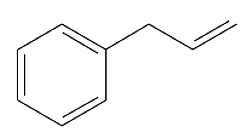
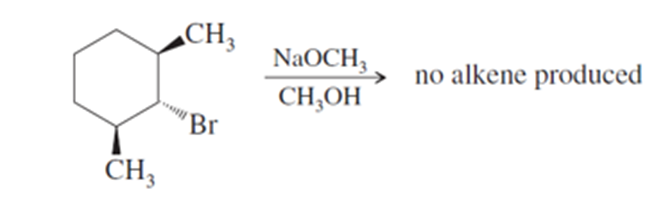
When the following stereoisomer of 2-bromo-1,3-dimethylcyclohexane is treated with sodium methoxide, no E2 reaction is observed. Explain why this compound cannot undergo the E2 reaction in the chair conformation.
Give the substitution and elimination products you would expect from the following reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.