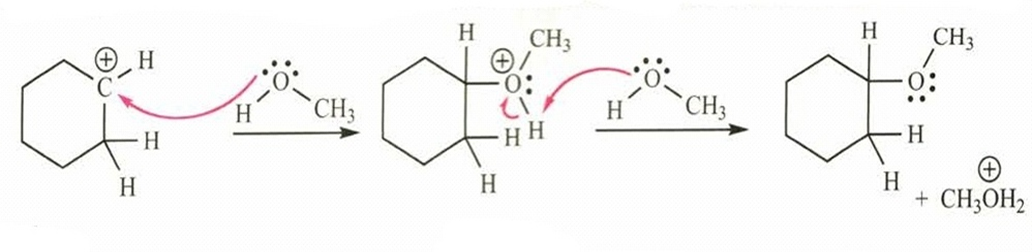
Chapter 7: 16P (page 356)
Show what happens in step-2 of the example if the solvent acts as a nucleophile (forming a bond to carbon) rather than as a base (removing a proton).
Short Answer
Learning Materials
Features
Discover
Chapter 7: 16P (page 356)
Show what happens in step-2 of the example if the solvent acts as a nucleophile (forming a bond to carbon) rather than as a base (removing a proton).

All the tools & learning materials you need for study success - in one app.
Get started for free
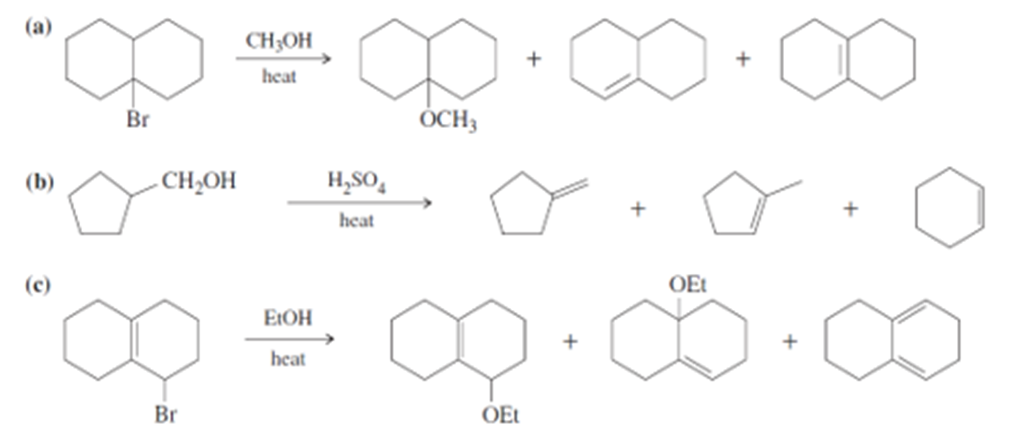
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown.
Predict the elimination products of the following reactions and label the major products.
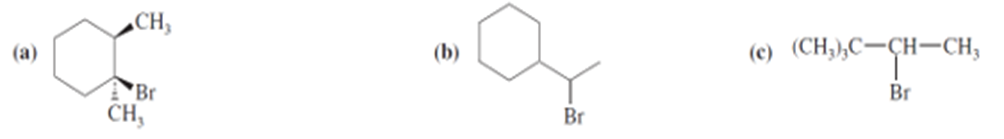
Predict the products of E1 elimination of the following compounds. Label the major products
Give the substitution and elimination products you would expect from the following reactions.
Propose mechanisms and draw reaction-energy diagrams for the following reactions. Pay particular attention to the structures of any transition states and intermediates. Compare the reaction-energy diagrams for the two reactions and explain the differences.
What do you think about this solution?
We value your feedback to improve our textbook solutions.