Chapter 1: 22P (page 80)
For each pair of structures, determine whether they represent different compounds or a single compound.
a.
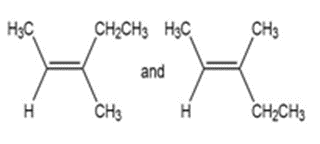
b.
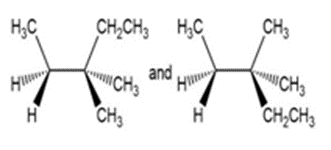
c,
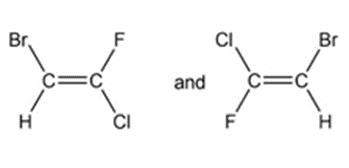
d.
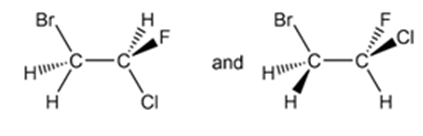
Short Answer
a) Different compounds
b) Identical compounds
c) Different compounds
d) Identical compounds
Learning Materials
Features
Discover
Chapter 1: 22P (page 80)
For each pair of structures, determine whether they represent different compounds or a single compound.
a.

b.

c,

d.

a) Different compounds
b) Identical compounds
c) Different compounds
d) Identical compounds
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw a line-angle formula for each compound in Problem.
For each of the following compounds,
1. Give the hybridization and approximate bond angles around each atom except hydrogen.
2. Draw a three-dimensional diagram, including any lone pairs of electrons.
(a) H3O+
(b) OH-
(c)CH2CHCN
(d)N(CH3)3
(e) CH3NH3+
(f) CH3COOH
(g) CH2CHNH
(h) CH3OH
(i) CH2O
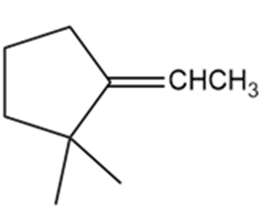
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.
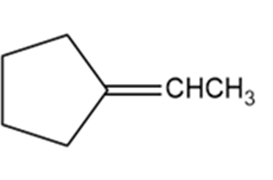
e.
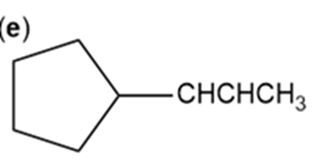
f.
Dimethyl sulfoxide (DMSO) has been used as an anti-inflammatory rub for race horses. DMSO and acetone appear to have similar structures, but thecarbon atom in acetone is planar, while thesulfur atom in DMSO is pyramidal. Draw Lewis structures for DMSO and acetone, predict the hybridization, and explain these observations.
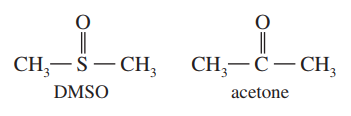
There is a small portion of the periodic table that you must know to do organic chemistry. Construct this part from memory, using the following steps.
a)From memory, make a list of the elements in the first two rows of the periodic table, together with their numbers of valence electrons.
b)Use this list to construct the first two rows of the periodic table.
c)Organic compounds often contain sulfur, phosphorus, chlorine, bromine and iodine. Add these elements to your periodic table.
What do you think about this solution?
We value your feedback to improve our textbook solutions.