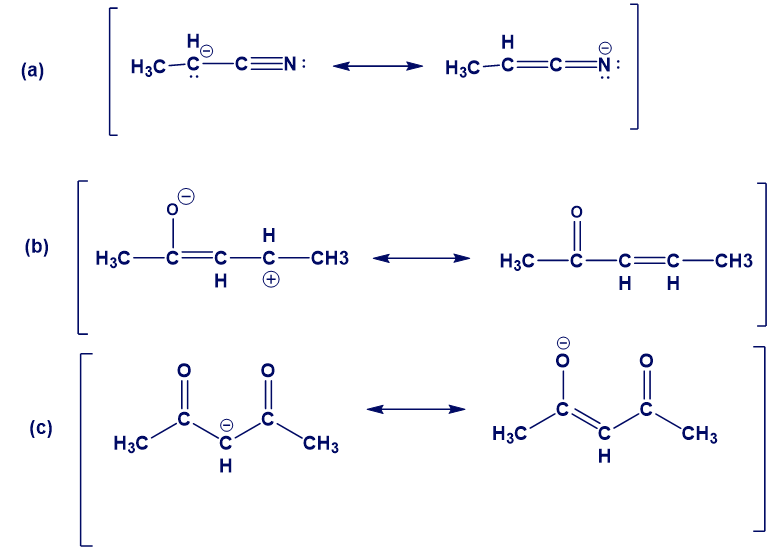
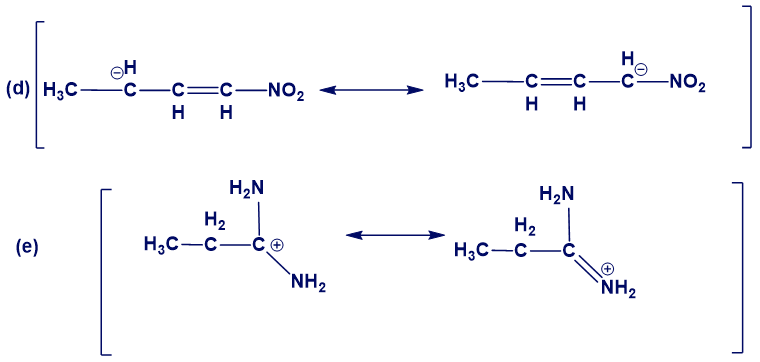
Chapter 1: 51P (page 89)
For each of the following compounds,
1. Give the hybridization and approximate bond angles around each atom except hydrogen.
2. Draw a three-dimensional diagram, including any lone pairs of electrons.
(a) H3O+
(b) OH-
(c)CH2CHCN
(d)N(CH3)3
(e) CH3NH3+
(f) CH3COOH
(g) CH2CHNH
(h) CH3OH
(i) CH2O
Short Answer
- The hybridization and bond angles around each atom accepts hydrogens.They are shown as below:
(a)
(b)
(c)
(d)
(e)
(f)
(g)
(h)
2. A three dimensional diagram is given there as well.