Chapter 1: 4P (page 46)
Circle any lone pair (pairs of nonbonding electrons) in the structures you drew for Problem1-3.
Short Answer
The compounds having unshared electrons pair are as follows:
a)
b)
c)
d)
e)
f)
g)
h)
i
j
k)
Learning Materials
Features
Discover
Chapter 1: 4P (page 46)
Circle any lone pair (pairs of nonbonding electrons) in the structures you drew for Problem1-3.
The compounds having unshared electrons pair are as follows:
a)
b)
c)
d)
e)
f)
g)
h)
i
j
k)
All the tools & learning materials you need for study success - in one app.
Get started for free
Name the element that corresponds to each electronic configuration.
Draw a Lewis structure for each compound. Include all nonbonding pairs of elctrons
(a CH3COCH2CHCHCOOH (b) NCCH2COCH2CHO
(c) CH2CHCH(OH)CH2CO2H (d) CH2CHC(CH3)CHCOOCH3
Allene,CH2=C=CH2 , has the structure shown below. Explain how the bonding in allene requires the two groups at its ends to be at right angles to each other.
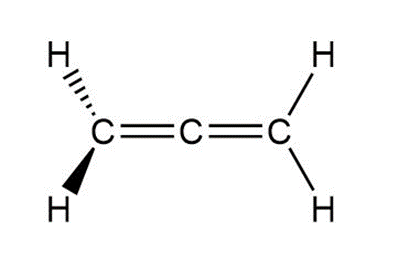
Allene
Give the molecular formula of each compound shown in Problem 1-37.
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic.
(a) KCl
(b) KOH
(c) CH3CH2Li
(d) CH3Cl
(e) KOCH3
(f) CH3CO2Na
(g) CCl4
What do you think about this solution?
We value your feedback to improve our textbook solutions.