Chapter 1: Q1-24P (page 82)
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.
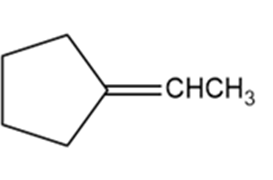
e.
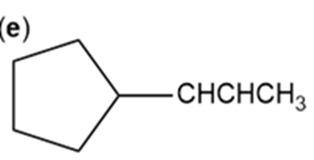
f.
Short Answer
Only compound (a) will show cis-trans isomerism.
Learning Materials
Features
Discover
Chapter 1: Q1-24P (page 82)
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.

e.

f.

Only compound (a) will show cis-trans isomerism.
All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethyl sulfoxide (DMSO) has been used as an anti-inflammatory rub for race horses. DMSO and acetone appear to have similar structures, but thecarbon atom in acetone is planar, while thesulfur atom in DMSO is pyramidal. Draw Lewis structures for DMSO and acetone, predict the hybridization, and explain these observations.
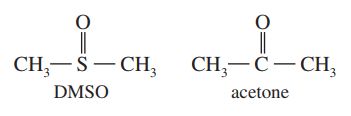
Question: Draw Lewis structure for the following compounds and ions, showing appropriate formal changes.
a. [CH3OH2]+
b. NH4Cl
c. (CH3)4NCl
d. NaOCH3
e. +CH3
f. -CH3
g. NaBH4
h. NaBH3CN
i. (CH3)2O-BF3
j. [HONH3]+
k. KOC(CH3)3
l. [H2C=OH]+
(a) Two compounds of formula C4H10
(b) Two compounds of formula C2H6O
(c) Two compounds of formula C2H7N
(d) Three compounds of formula C2H7NO
(e) Three compounds of formula C3H8O2
(f) Three compounds of formula C2H4O
(a) Use your molecular models to make thane, and compare the model with the preceding structures. (b) Make a model of butane (C4H10), and draw this model using dashed lines and wedges to represent bonds going back and coming forward.
Question: Draw a Lewis structure for each species.
What do you think about this solution?
We value your feedback to improve our textbook solutions.