Chapter 1: Q1-22P (page 80)
For each pair of structures, determine whether they represent different compounds or a single compound.
a.
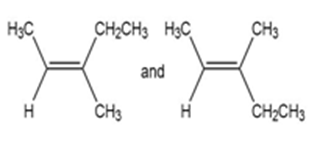
b.
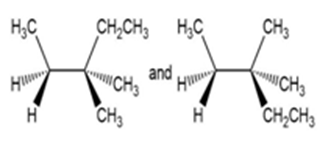
c.
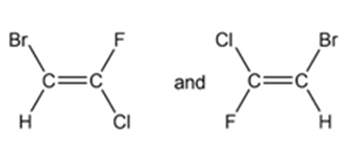
d.
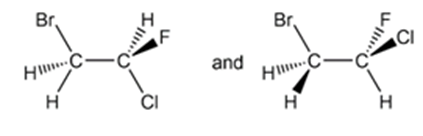
Short Answer
- Different compounds
- Identical compounds
- Different compounds
- Identical compounds
Learning Materials
Features
Discover
Chapter 1: Q1-22P (page 80)
For each pair of structures, determine whether they represent different compounds or a single compound.
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many hydrogens are present around each carbon atom in the following molecules?
a. 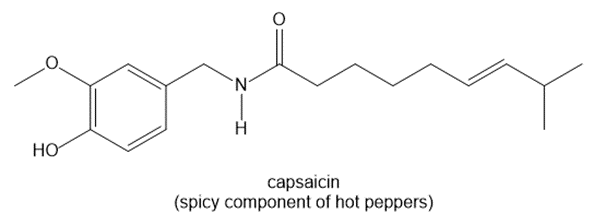
b.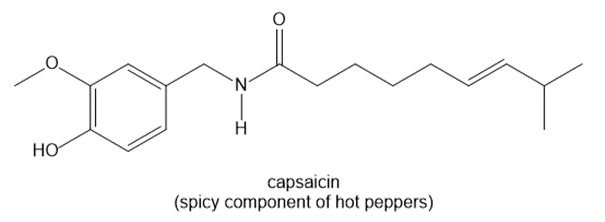
In pent− 2 −yne (CH3CCCH2CH3), there are four atoms in a straight line. Use dashed lines and wedges to draw three-dimensional representation of this molecule, and circle the four atoms that are in a straight line.
In 1934, Edward A. Doisy of Washington University extracted 3000 lb of hog ovaries to isolate a few milligrams of pure estradiol, a potent female hormone. Doisy burned 5.00 mg of this precious sample in oxygen and found that 14.54 mg of CO2 and 3.97 mg of H2O were generated.
(a) Determine the empirical formula of estradiol.
(b) The molecular weight of estradiol was later determined to be 272. Determine the molecular formula of estradiol.
(a) Predict the hybridization of the oxygen atom in water H2O,. Draw a picture of its three-dimensional structure, and explain why its bond angle is 104.50.
(b) The electrostatic potential maps for ammonia and water are shown here. The structure of ammonia is shown within its EPM. Note how the lone pair creates a region of high electron potential (red), and the hydrogens are in regions of low electron potential (blue). Show how your three-dimensional structure of water corresponds with its EPM.

(a) Two compounds of formula C4H10
(b) Two compounds of formula C2H6O
(c) Two compounds of formula C2H7N
(d) Three compounds of formula C2H7NO
(e) Three compounds of formula C3H8O2
(f) Three compounds of formula C2H4O
What do you think about this solution?
We value your feedback to improve our textbook solutions.