Chapter 1: Q59. (page 56)
Question: How many hydrogens are present around each carbon atom in the following molecules?
a. 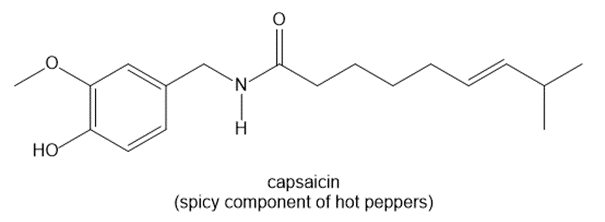
b.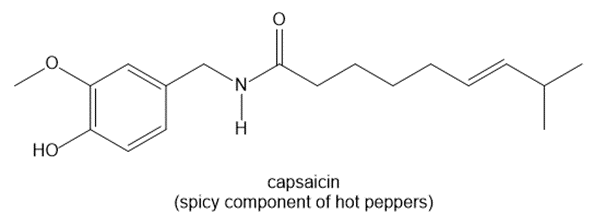
Short Answer
Answer
a. The number of hydrogen atoms is 27.
b. The number of hydrogen atoms is 39.
Learning Materials
Features
Discover
Chapter 1: Q59. (page 56)
Question: How many hydrogens are present around each carbon atom in the following molecules?
a. 
b.
Answer
a. The number of hydrogen atoms is 27.
b. The number of hydrogen atoms is 39.
All the tools & learning materials you need for study success - in one app.
Get started for free
For each compound, state whether its bonding is covalent, ionic, or a mixture of covalent and ionic.
(a) KCl
(b) KOH
(c)
(d)
(e)
(f)
(g)
Circle any lone pair (pairs of nonbonding electrons) in the structures you drew for Problem1-3.
Give the molecular formula of each compound shown in Problem 1-37.
In pent− 2 −yne (CH3CCCH2CH3), there are four atoms in a straight line. Use dashed lines and wedges to draw three-dimensional representation of this molecule, and circle the four atoms that are in a straight line.
Name the element that corresponds to each electronic configuration
a) 1s22s22p63s1
b)1s22s22p6
c)1s22s22p1
d)1s22s22p63s2
What do you think about this solution?
We value your feedback to improve our textbook solutions.