Chapter 1: 11P (page 60)

Short Answer
The structures are as follows.
Learning Materials
Features
Discover
Chapter 1: 11P (page 60)

The structures are as follows.
All the tools & learning materials you need for study success - in one app.
Get started for free
In the following sets of resonance forms, label the major and minor contributors and state which structures would be of equal energy. Add any missing resonance forms.
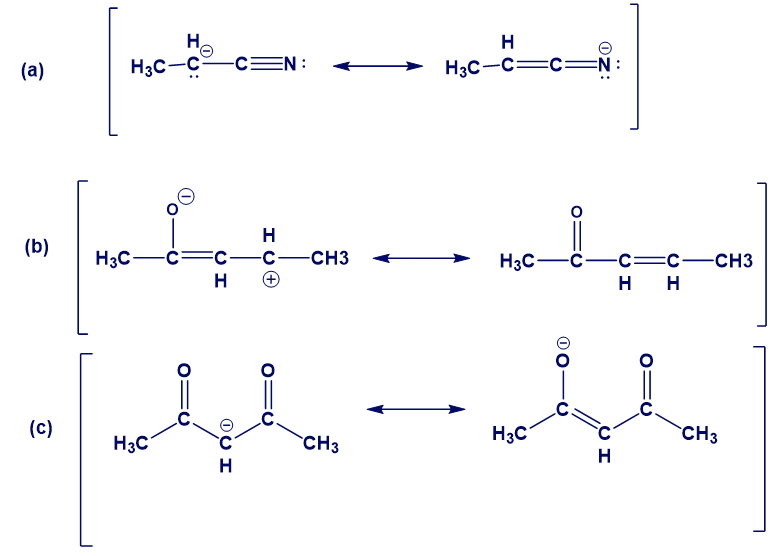
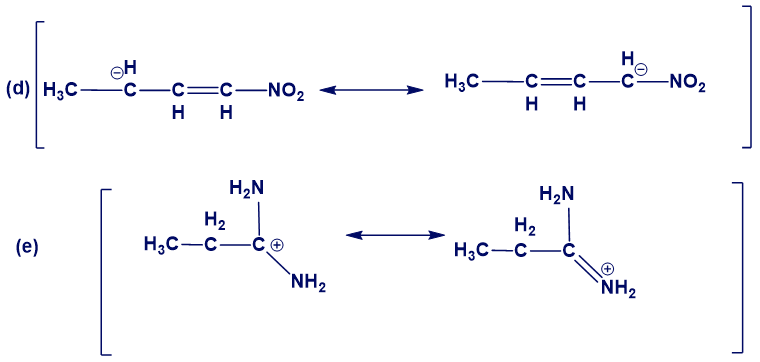
Question: How many hydrogens are present around each carbon atom in the following molecules?
a. 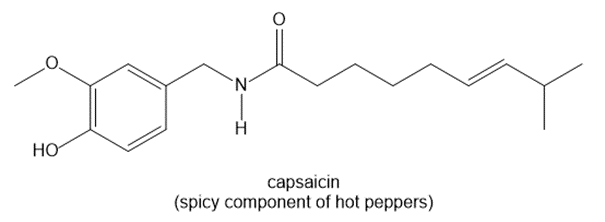
b.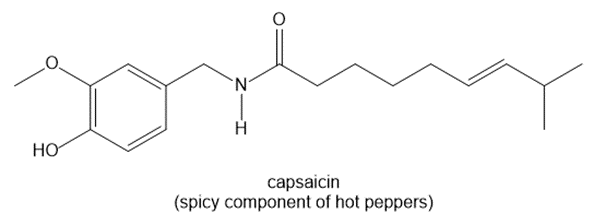
Predict the hybridization, geometry, and bond angles for the carbon and nitrogen atoms in acetonitriles
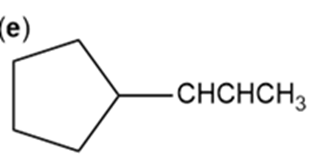
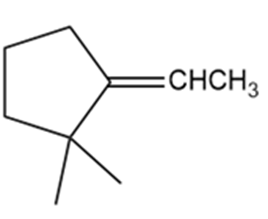
Give Lewis structures corresponding to the following line-angle structures. Give the molecular formula for each structure.
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of those that do.
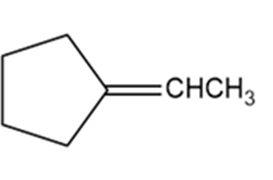
e.
f.
What do you think about this solution?
We value your feedback to improve our textbook solutions.