Chapter 1: 12P (page 61)
Give Lewis structures corresponding to the following line-angle structures. Give the molecular formula for each structure.
Short Answer
a.
b.
c.
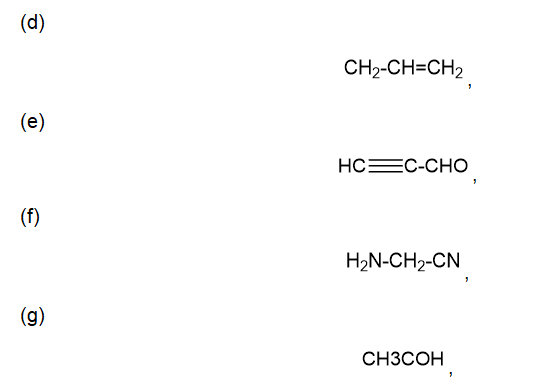
d.
e
f
g.
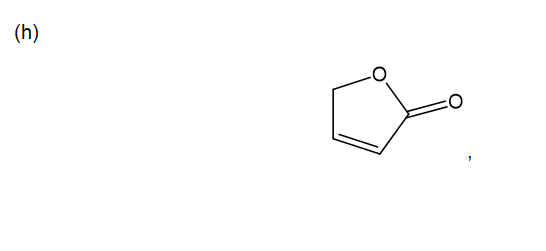
h
Learning Materials
Features
Discover
Chapter 1: 12P (page 61)
Give Lewis structures corresponding to the following line-angle structures. Give the molecular formula for each structure.
a.
b.
c.
d.
e
f
g.
h
All the tools & learning materials you need for study success - in one app.
Get started for free
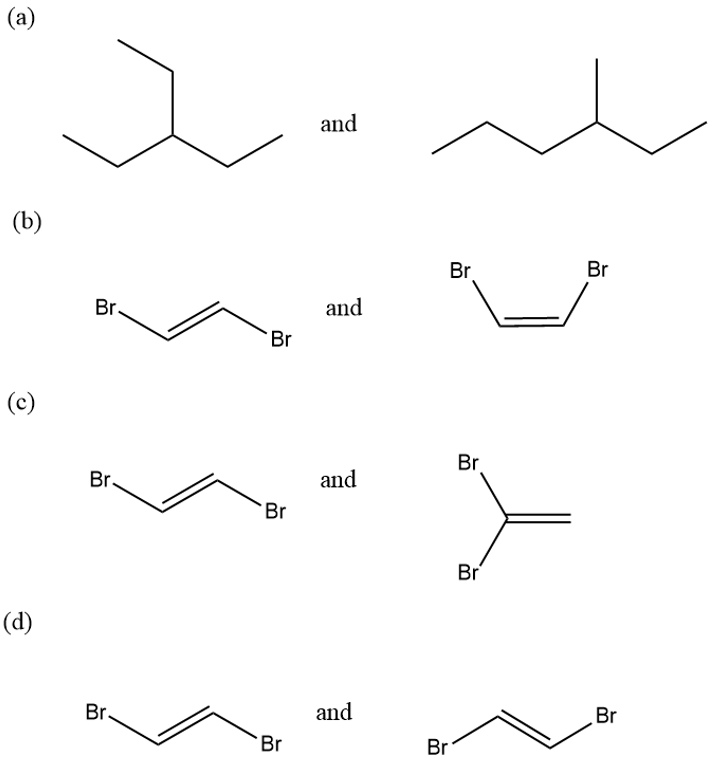
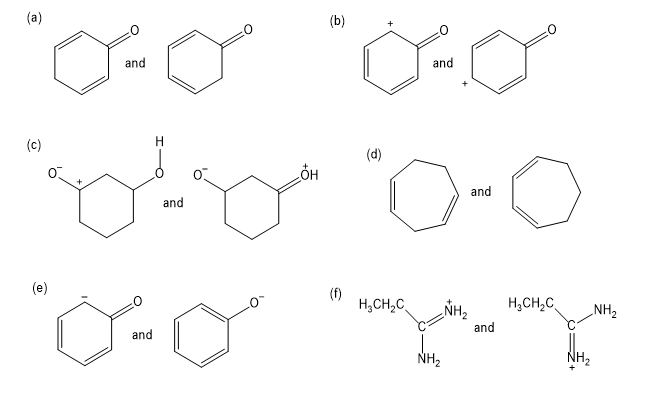
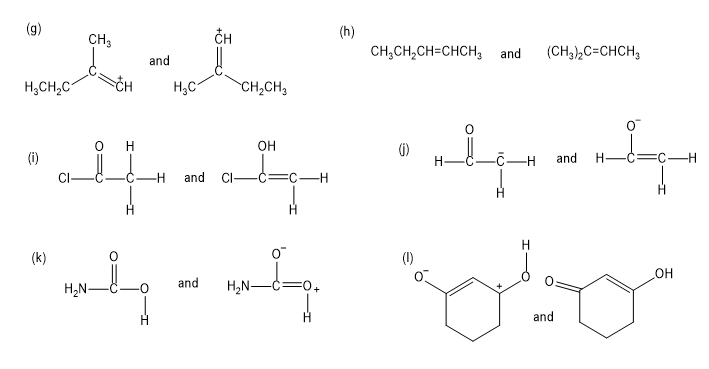
Give the relationship between the following pairs of structures. The relationships are:
Same compound
Constitutional isomers (structural isomers)
Cis-trans isomers
Not isomers (different molecular formula)
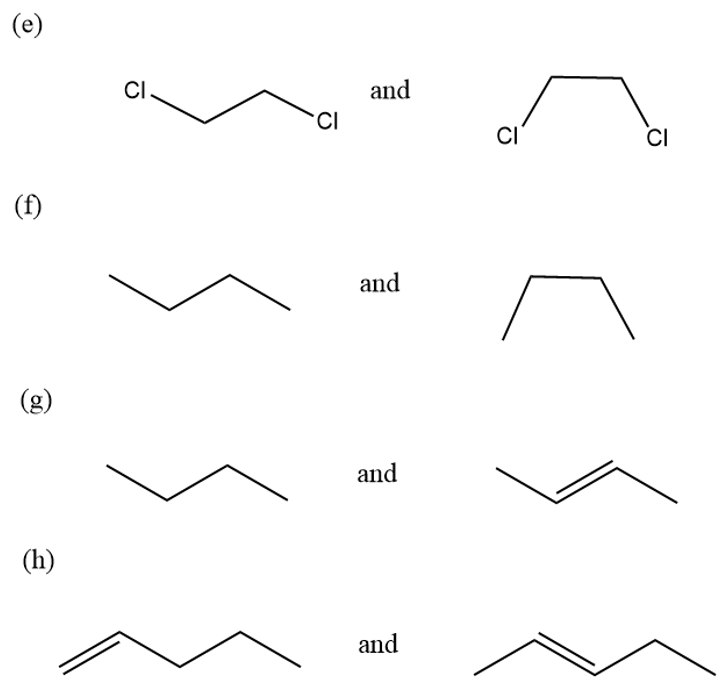
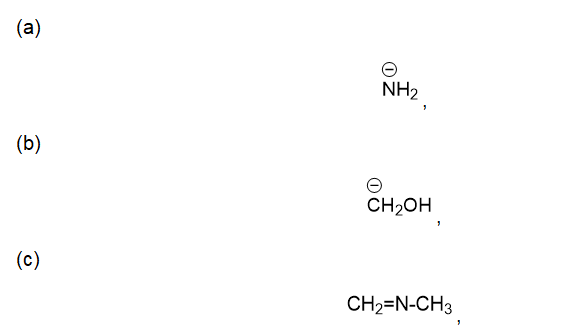
For each of the following compounds and ions,
1. Draw a Lewis structure.
2. Show the kinds of orbitals that overlap to form each bond.
3. Give approximate bond angles around each atom except hydrogen.

Draw a Lewis structure for each compound. Include all nonbonding pairs of elctrons
(a CH3COCH2CHCHCOOH (b) NCCH2COCH2CHO
(c) CH2CHCH(OH)CH2CO2H (d) CH2CHC(CH3)CHCOOCH3
Determine whether the following pairs of structures are different compounds or simply resonance forms of the same compounds.
Draw a complete Structural formula and a condensed structural formula for
(a) three compounds of formula C3H8O
(b) five compound of formula C3H6O
What do you think about this solution?
We value your feedback to improve our textbook solutions.