Chapter 17: Q34P (page 890)
Propose a mechanism for the bromination of ethyl benzene shown above.
Short Answer
Initiation:
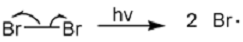
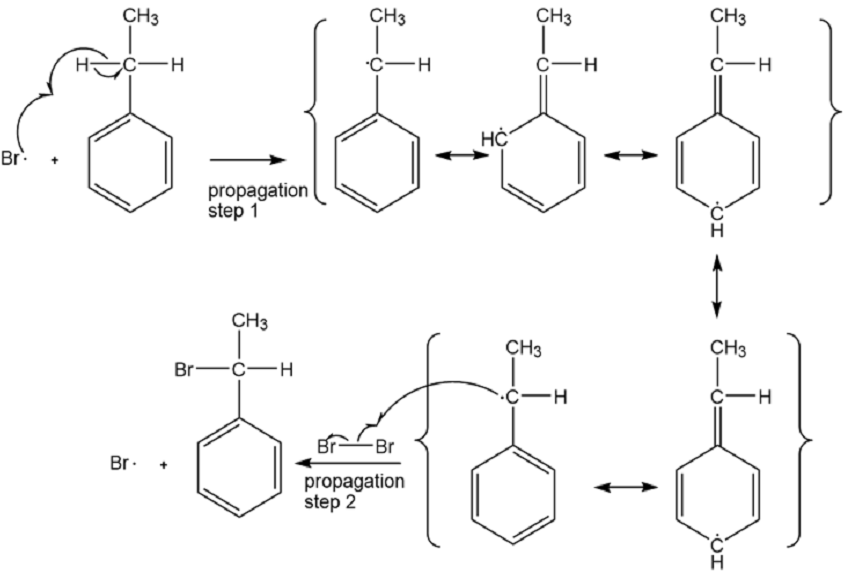
Propagation:
Learning Materials
Features
Discover
Chapter 17: Q34P (page 890)
Propose a mechanism for the bromination of ethyl benzene shown above.
Initiation:

Propagation:

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize the following compounds, using the indicated starting materials.
(a) 3-phenylbutan-1-ol from styrene
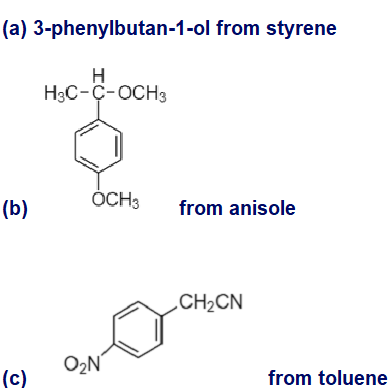
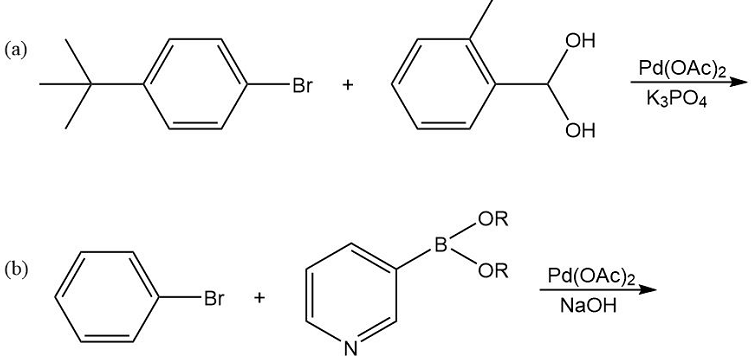
What products would you expect from the following Suzuki coupling reactions?
Predict the major products of the following reactions.
(a) toluene + excess CI2 (heat, pressure)
(b) benzamide ( PhCONH2 ) + Na (liquid NH3, CH3 CH2OH)
(c) o-xylene + H2 (1000 psi, 100 °C, Rh catalyst)
(d) p-xylene + Na (liquid NH3, CH3CH2OH )
(e) 
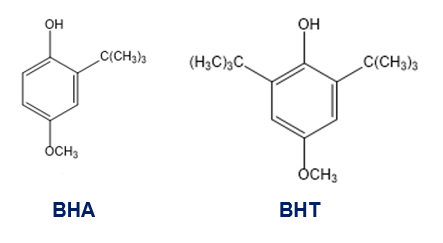
The antioxidants BHA and BHT are commonly used as food preservatives. Show how BHA and BHT can be made from phenol and hydroquinone.
The bombardier beetle defends itself by spraying a hot quinone solution from its abdomen (see photo). This solution is formed by the enzyme-catalyzed oxidation of hydroquinone by hydrogen peroxide. Write a balanced equation for this oxidation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.