Chapter 17: Q36P (page 890)
Predict the major products when the following compounds are irradiated by light and treated with (1) 1 equivalent of Br2 and (2) excess Br2.
(a) isopropylbenzene
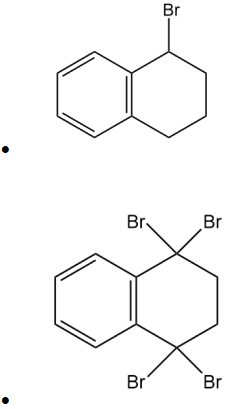
(b) tetralin
Short Answer
(a) isopropylbenzene

(b) tetralin
Learning Materials
Features
Discover
Chapter 17: Q36P (page 890)
Predict the major products when the following compounds are irradiated by light and treated with (1) 1 equivalent of Br2 and (2) excess Br2.
(a) isopropylbenzene
(b) tetralin
(a) isopropylbenzene

(b) tetralin

All the tools & learning materials you need for study success - in one app.
Get started for free
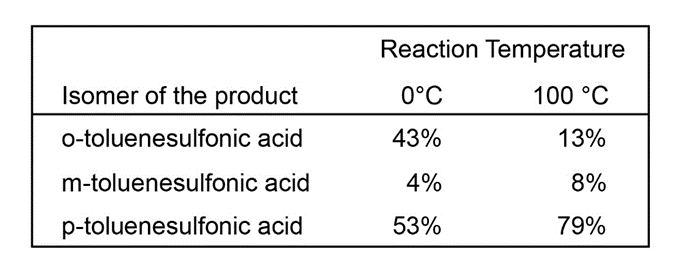
Unlike most other electrophilic aromatic substitutions, sulfonation is often reversible. When one sample of toluene is sulfonated at 0 °C and another sample is sulfonated at 100 °C, the following ratios of substitution products result:
(a) Explain the change in the product ratios when the temperature is increased.
(b) Predict what will happen when the product mixture from the reaction at 0 °C is heated to 100 °C.
(c) Because the SO3Hgroup can be added to a benzene ring and removed later, it is sometimes called a blocking group. Show how 2,6-dibromotoluene can be made from toluene using sulfonation and desulfonation as intermediate steps in the synthesis
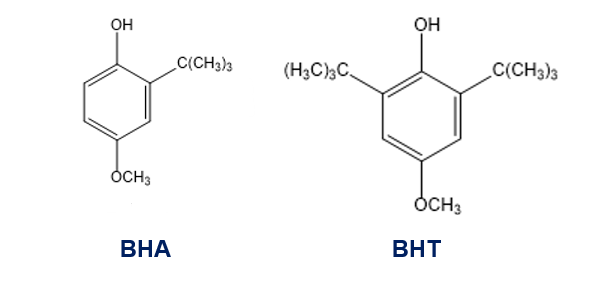
The antioxidants BHA and BHT are commonly used as food preservatives. Show how BHA and BHT can be made from phenol and hydroquinone.
Indane can undergo free-radical chlorination at any of the alkyl positions on the aliphatic ring.
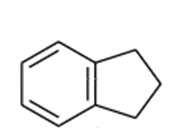
indane
Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species, and explain this unusual behavior.
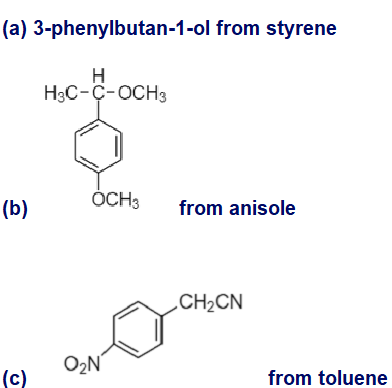
Show how you would synthesize the following compounds, using the indicated starting materials.
(a) 3-phenylbutan-1-ol from styrene
What do you think about this solution?
We value your feedback to improve our textbook solutions.