Chapter 17: Q37P (page 891)
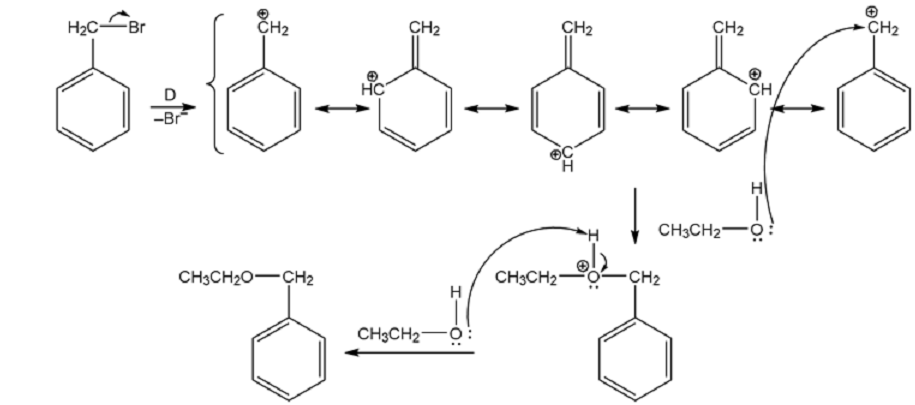
Propose a mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether (shown above).
Short Answer
The mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether.
Learning Materials
Features
Discover
Chapter 17: Q37P (page 891)
Propose a mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether (shown above).
The mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether.

All the tools & learning materials you need for study success - in one app.
Get started for free
Phenolphthalein, a common nonprescription laxative, is also an acid-base indicator that is colorless in acid and red in base. Phenolphthalein is synthesized by the acid-catalyzed reaction of phthalic anhydride with 2 equivalents of phenol.
(a) Propose a mechanism for the synthesis of phenolphthalein.
(b) Propose a mechanism for the conversion of phenolphthalein to its red dianion in the base.
(c) Use resonance structures to show that the two phenolic oxygen atoms are equivalent (each with half a negative charge) in the red phenolphthalein dianion.
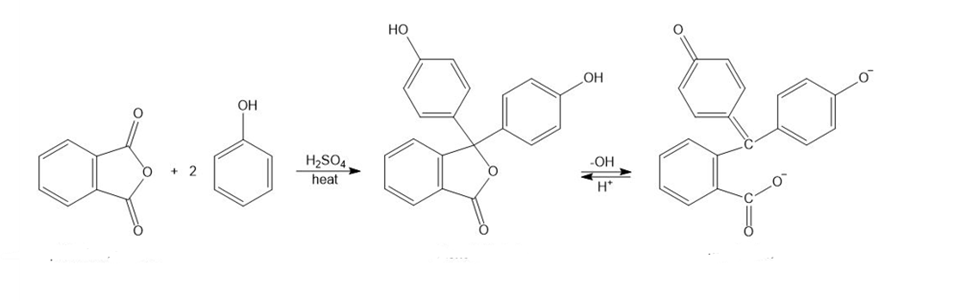
Phthalic anhydride Phenolphthalein red dianion
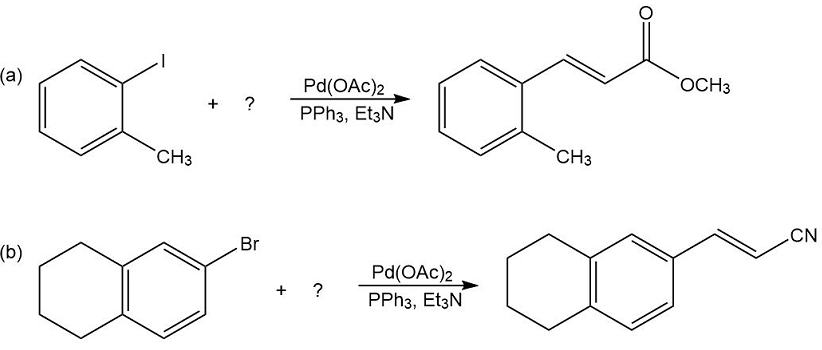
What products would you expect from the following Suzuki coupling reactions?

To synthesize m-ethylbenzenesulfonic acid, a student attempted the Friedel–Crafts alkylation of benzenesulfonic acid with bromoethane. Do you predict that this reaction was successful? If not, propose an alternative synthesis.
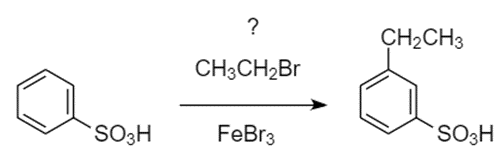
m-ethylbenzenesulfonic acid
Phenol reacts with three equivalents of bromine in CCl4(in the dark) to give a product of formula C6H3OBr3. When this product is added to bromine water, a yellow solid of molecular formula C6H2OBr4 precipitates out of the solution. The IR spectrum of the yellow precipitate shows a strong absorption (much like that of a quinone) around 1680cm-1 . Propose structures for the two products.
What substituted alkene would you use in the Heck reaction to make the following products?
What do you think about this solution?
We value your feedback to improve our textbook solutions.