Chapter 17: Q38P (page 892)
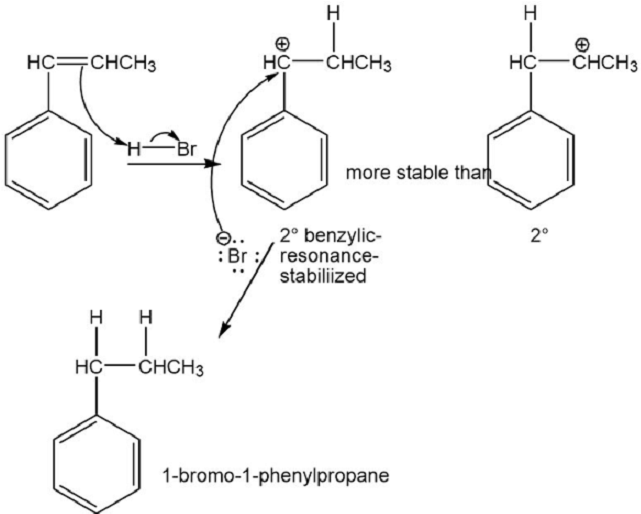
(a) Based on what you know about the relative stabilities of alkyl cations and benzylic cations, predict the product of addition of HBr to 1-phenylpropene.
(b) Propose a mechanism for this reaction.
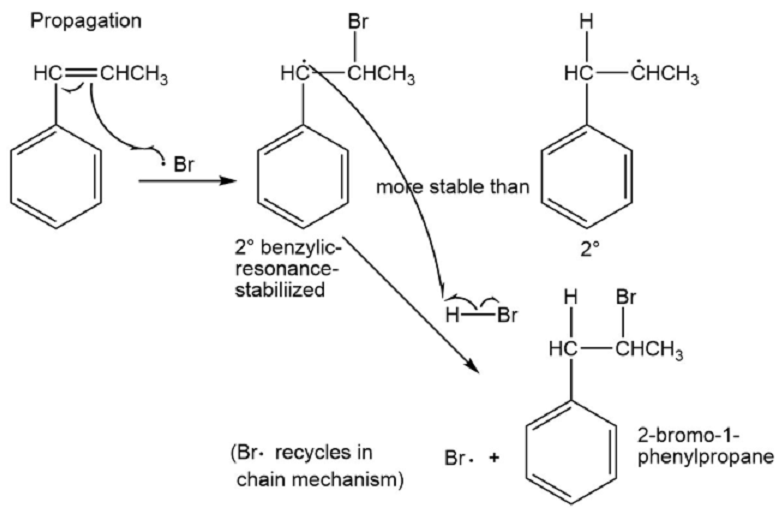
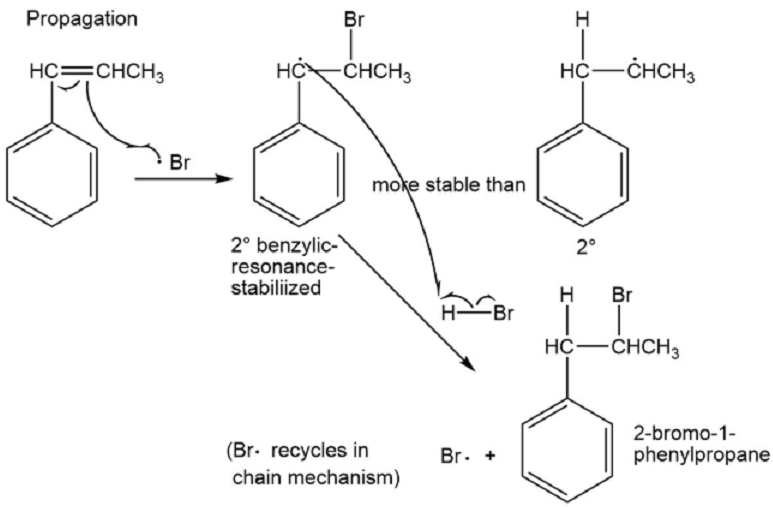
(c) Based on what you know about the relative stabilities of alkyl radicals and benzylic radicals, predict the product of addition of HBr to 1-phenylpropene in the presence of a free-radical initiator.
(d) Propose a mechanism for this reaction.
Short Answer
⦁ Benzylic cations are more stable than alkyl cations because of resonance. The product of the addition of HBr to 1-phenylpropene is 1-bromo-1-phenylpropane.
⦁ The mechanism:
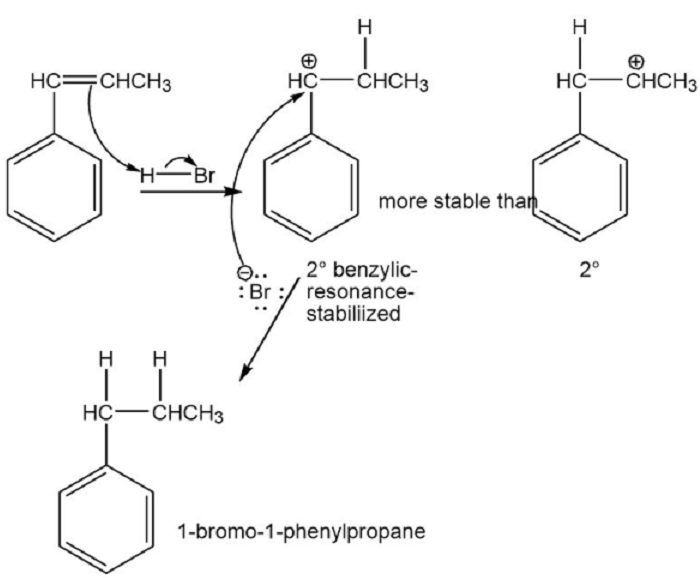
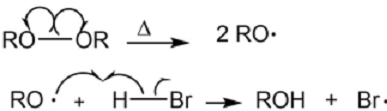
⦁ Benzylic radicals are more stable than alkyl radicals because of resonance. The product of the addition of HBr to 1-phenylpropene in presence of a free radical initiator is 2-bromo-1-phenylpropane.
⦁ The mechanism: