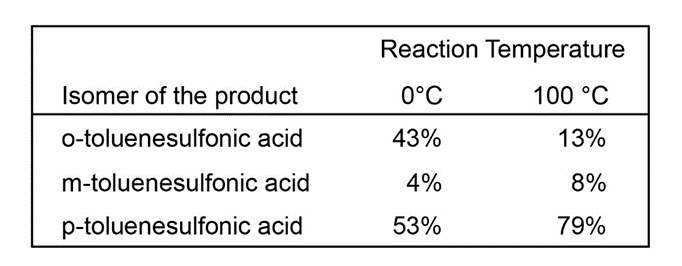
Unlike most other electrophilic aromatic substitutions, sulfonation is often reversible. When one sample of toluene is sulfonated at 0 °C and another sample is sulfonated at 100 °C, the following ratios of substitution products result:
(a) Explain the change in the product ratios when the temperature is increased.
(b) Predict what will happen when the product mixture from the reaction at 0 °C is heated to 100 °C.
(c) Because the SO3Hgroup can be added to a benzene ring and removed later, it is sometimes called a blocking group. Show how 2,6-dibromotoluene can be made from toluene using sulfonation and desulfonation as intermediate steps in the synthesis

 Formation of acetophenone
Formation of acetophenone Formation of 3-chloro acetophenone
Formation of 3-chloro acetophenone Formation of the desired product
Formation of the desired product Formation of acetophenone
Formation of acetophenone Formation of 3-chloro acetophenone
Formation of 3-chloro acetophenone Formation of the desired product
Formation of the desired product



