Chapter 17: Q73P (page 910)
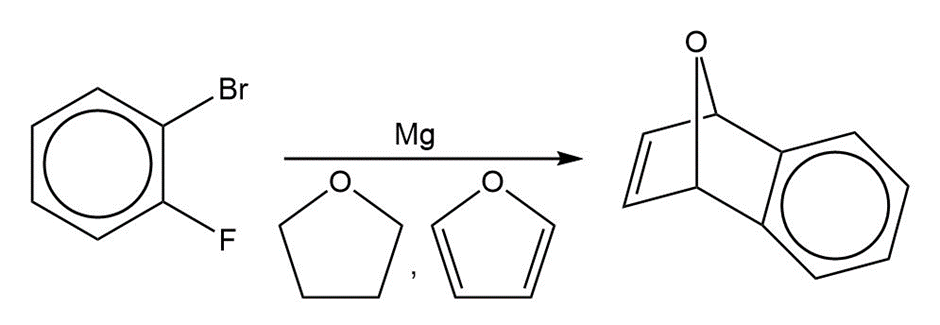
A graduate student tried to make o-fluorophenylmagnesium bromide by adding magnesium to an ether solution of o-fluorobromobenzene. After obtaining puzzling results with this reaction, she repeated the reaction by using as solvent some tetrahydrofuran that contained a small amount of furan. From this reaction, she isolated a fair yield of the compound that follows. Propose a mechanism for its formation
Short Answer
 Mechanism of the reaction
Mechanism of the reaction
 Formation of Diels alder adduct
Formation of Diels alder adduct