Chapter 17: Q2P (page 849)
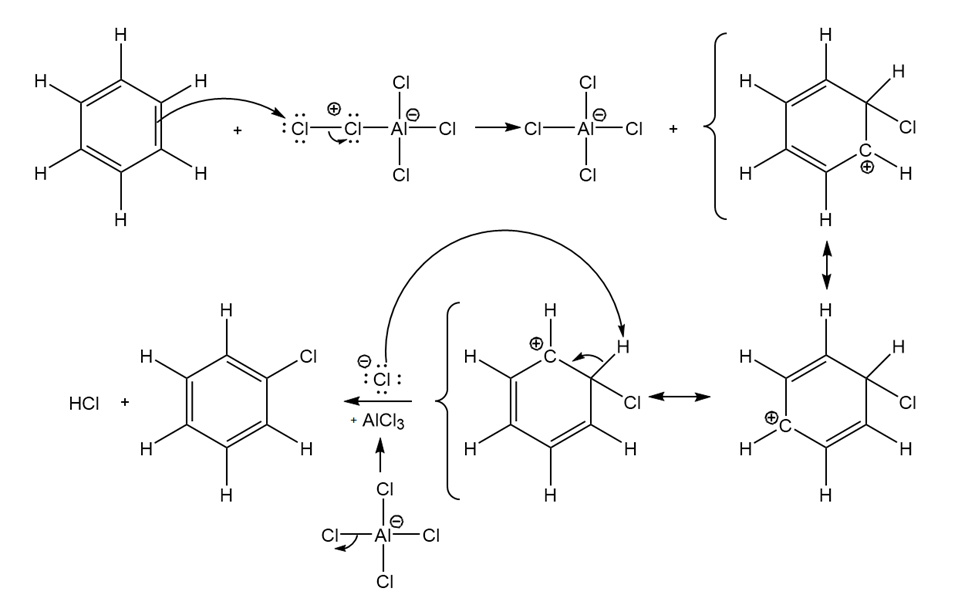
Propose a mechanism for the aluminum chloride-catalyzed reaction of benzene with chlorine.
Short Answer
The mechanism for the aluminum chloride-catalyzed reaction of benzene with chlorine is as follows:
Learning Materials
Features
Discover
Chapter 17: Q2P (page 849)
Propose a mechanism for the aluminum chloride-catalyzed reaction of benzene with chlorine.
The mechanism for the aluminum chloride-catalyzed reaction of benzene with chlorine is as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
The highly reactive triple bond of benzyne is a powerful dienophile. Predict the product of the Diels–Alder reaction of benzyne (from chlorobenzene and NaOH, heated) with cyclopentadiene.
Phenolphthalein, a common nonprescription laxative, is also an acid-base indicator that is colorless in acid and red in base. Phenolphthalein is synthesized by the acid-catalyzed reaction of phthalic anhydride with 2 equivalents of phenol.
(a) Propose a mechanism for the synthesis of phenolphthalein.
(b) Propose a mechanism for the conversion of phenolphthalein to its red dianion in the base.
(c) Use resonance structures to show that the two phenolic oxygen atoms are equivalent (each with half a negative charge) in the red phenolphthalein dianion.
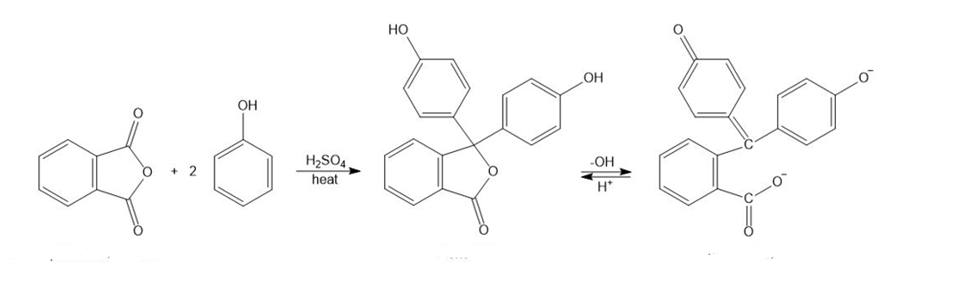
Phthalic anhydride Phenolphthalein red dianion
Step 2 of the iodination of benzene shows water acting as a base and removing a proton from the sigma complex. We did not consider the possibility of water acting as a nucleophile and attacking the carbocation, as in an electrophilic addition to an alkene. Draw the reaction that would occur if water reacted as a nucleophile and added to the carbocation. Explain why this type of addition is rarely observed.
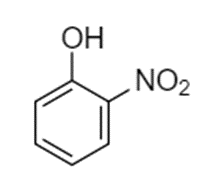
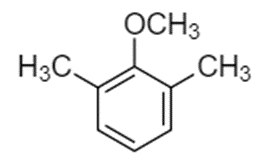
Predict the site(s) of electrophilic attack on these compounds.
(a)
(b)
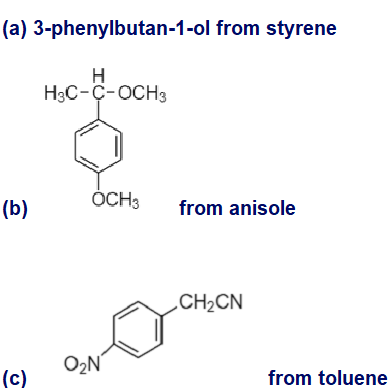
Show how you would synthesize the following compounds, using the indicated starting materials.
(a) 3-phenylbutan-1-ol from styrene
What do you think about this solution?
We value your feedback to improve our textbook solutions.