Chapter 17: Q29P (page 885)
What products would you expect from the following Suzuki coupling reactions?
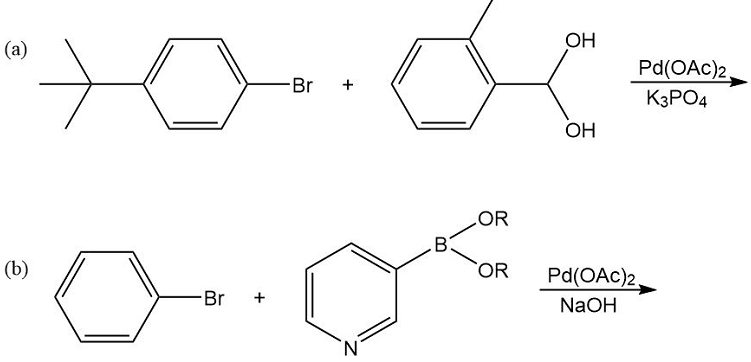
Short Answer
⦁ The product expected from the first reaction:
⦁ The product expected from the second reaction:
Learning Materials
Features
Discover
Chapter 17: Q29P (page 885)
What products would you expect from the following Suzuki coupling reactions?

⦁ The product expected from the first reaction:
⦁ The product expected from the second reaction:

All the tools & learning materials you need for study success - in one app.
Get started for free
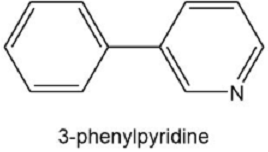
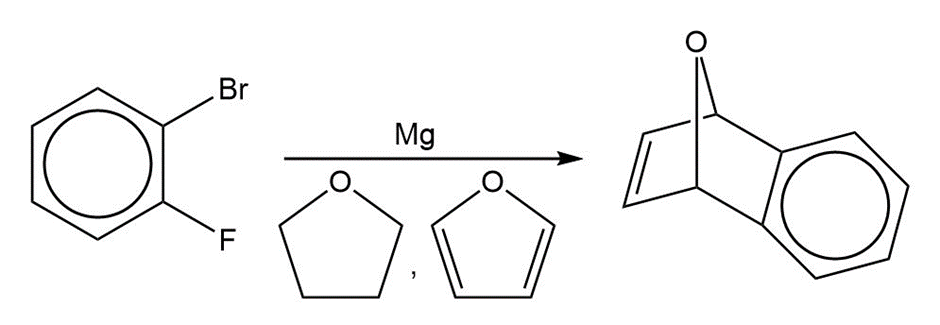
A graduate student tried to make o-fluorophenylmagnesium bromide by adding magnesium to an ether solution of o-fluorobromobenzene. After obtaining puzzling results with this reaction, she repeated the reaction by using as solvent some tetrahydrofuran that contained a small amount of furan. From this reaction, she isolated a fair yield of the compound that follows. Propose a mechanism for its formation
What products would you expect from the following reactions?

Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species, and explain this unusual behavior.
Phenolphthalein, a common nonprescription laxative, is also an acid-base indicator that is colorless in acid and red in base. Phenolphthalein is synthesized by the acid-catalyzed reaction of phthalic anhydride with 2 equivalents of phenol.
(a) Propose a mechanism for the synthesis of phenolphthalein.
(b) Propose a mechanism for the conversion of phenolphthalein to its red dianion in the base.
(c) Use resonance structures to show that the two phenolic oxygen atoms are equivalent (each with half a negative charge) in the red phenolphthalein dianion.
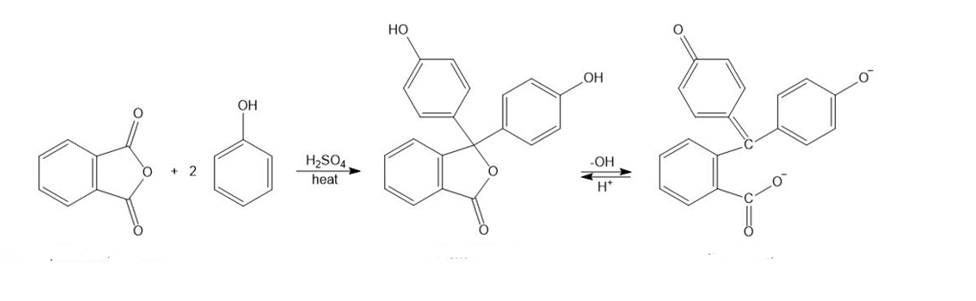
Phthalic anhydride Phenolphthalein red dianion
Propose mechanisms for the Birch reductions of benzoic acid and anisole just shown. Show why the observed orientation of reduction is favored in each case.
What do you think about this solution?
We value your feedback to improve our textbook solutions.