Chapter 17: 77P (page 845)
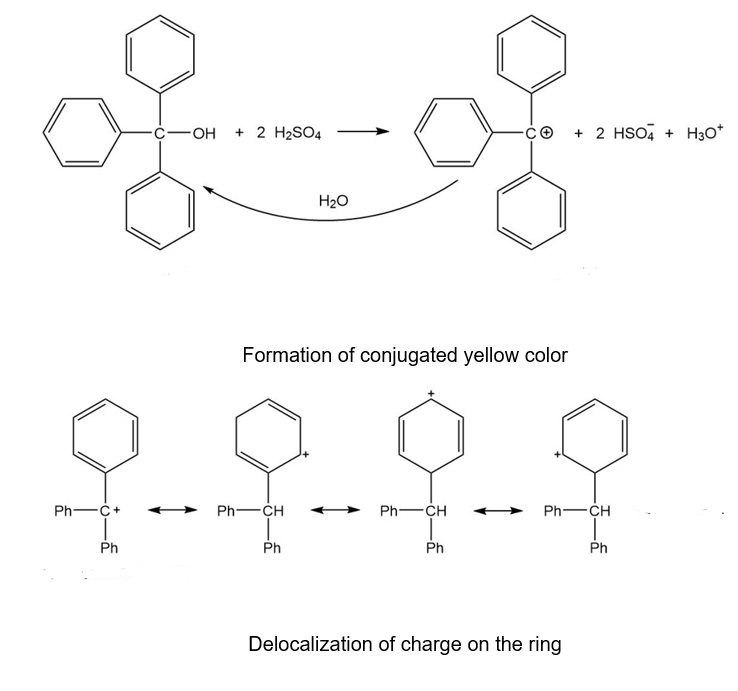
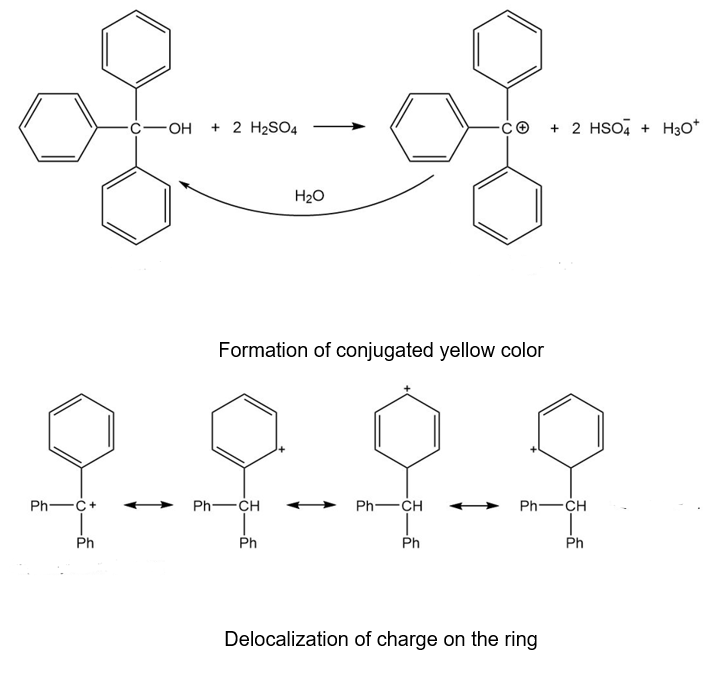
Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species, and explain this unusual behavior.
Short Answer