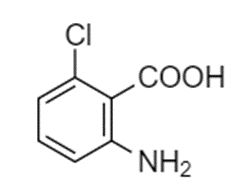
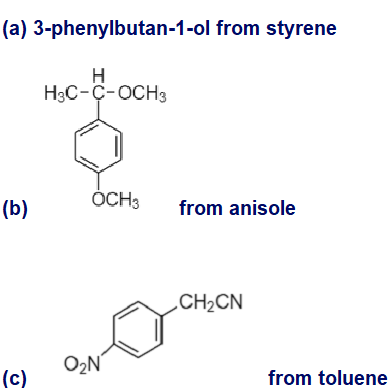
Chapter 17: Q40P (page 893)
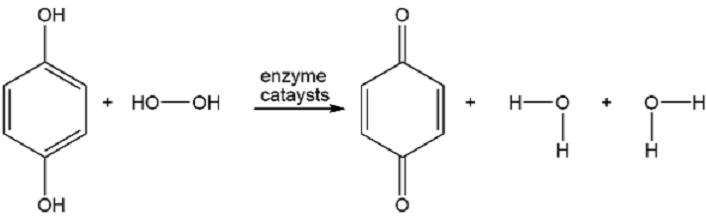
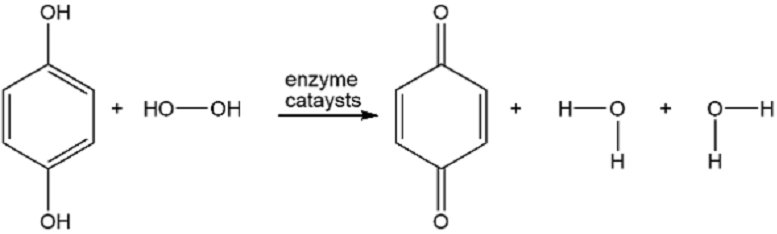
The bombardier beetle defends itself by spraying a hot quinone solution from its abdomen (see photo). This solution is formed by the enzyme-catalyzed oxidation of hydroquinone by hydrogen peroxide. Write a balanced equation for this oxidation.
Short Answer