Chapter 17: Q49P (page 899)
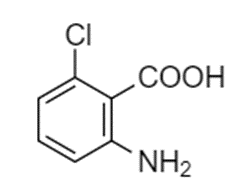
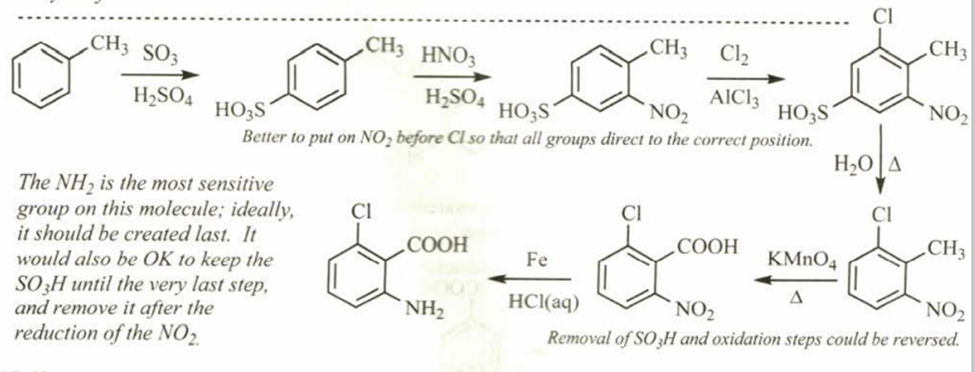
Starting from toluene, propose a synthesis of this trisubstituted benzene
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q49P (page 899)
Starting from toluene, propose a synthesis of this trisubstituted benzene


All the tools & learning materials you need for study success - in one app.
Get started for free
When anthracene is added to the reaction of chlorobenzene with concentrated NaOH at 350 °C, an interesting Diels–Alder adduct of the formula C20H14results. The proton NMR spectrum of the product shows a singlet of area 2 around d 3 and a broad singlet of area 12 around d 7. Propose a structure for the product, and explain why one of the aromatic rings of anthracene reacted as a diene.
Triphenylmethanol is insoluble in water, but when it is treated with concentrated sulfuric acid, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species, and explain this unusual behavior.
Predict the major products of treating the following compounds with hot, concentrated potassium permanganate, followed by acidification with dilute HCl.
(a) isopropylbenzene
(b) p-xylene
(c) tetralin
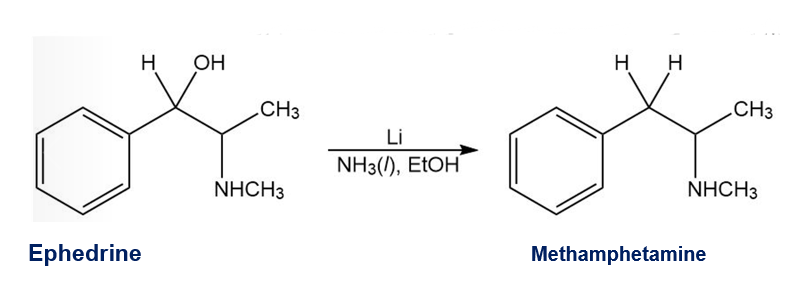
A common illicit synthesis of methamphetamine involves an interesting variation of the Birch reduction. A solution of ephedrine in alcohol is added to liquid ammonia, followed by several pieces of lithium metal. The Birch reduction usually reduces the aromatic ring, but in this case, it eliminates the hydroxy group of ephedrine to give methamphetamine. Propose a mechanism, similar to that for the Birch reduction, to explain this unusual course of the reaction
Indane can undergo free-radical chlorination at any of the alkyl positions on the aliphatic ring.
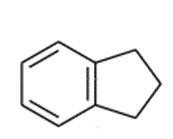
indane
What do you think about this solution?
We value your feedback to improve our textbook solutions.