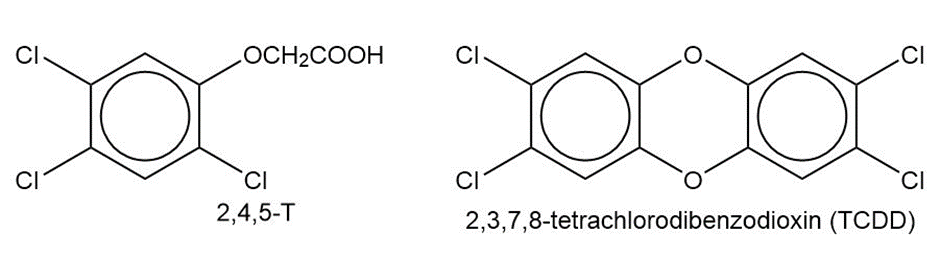
In Chapter 14, we saw that Agent Orange contains (2,4,5-trichlorophenoxy) acetic acid, called 2,4,5-T. This compound is synthesized by the partial reaction of 1,2,4,5-tetrachlorobenzene with sodium hydroxide, followed by a reaction with sodium chloroacetate, ClCH2CO2Na.
(a) Draw the structures of these compounds, and write equations for these reactions.
(b) One of the impurities in the Agent Orange used in Vietnam was 2,3,7,8-tetrachlorodibenzodioxin (2,3,7,8-TCDD), often incorrectly called “dioxin.” Propose a mechanism to show how 2,3,7,8-TCDD is formed in the synthesis of 2,4,5-T.
(c) Show how the TCDD contamination might be eliminated, both after the first step and on completion of the synthesis