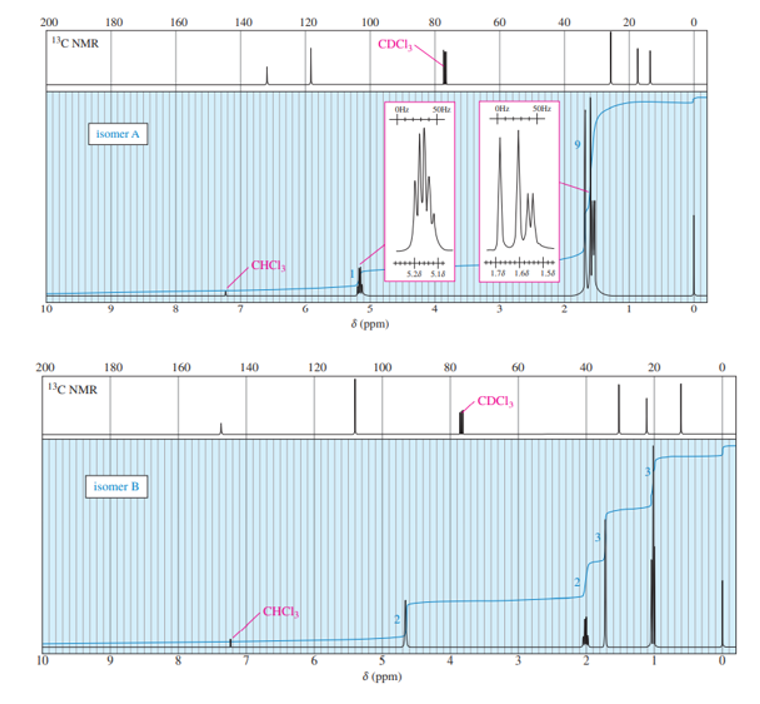
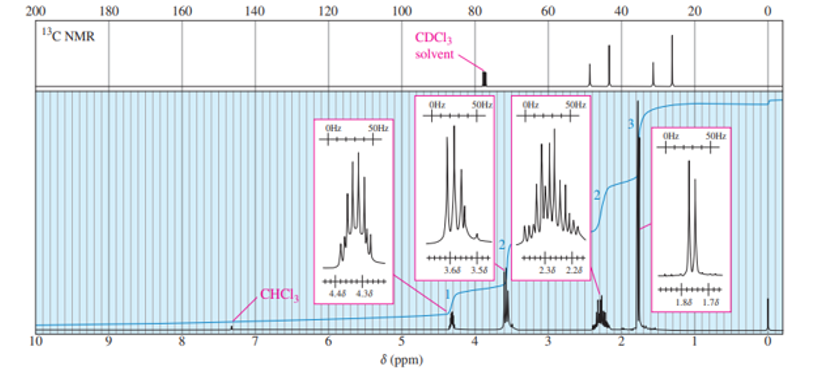
Chapter 13: Q28P (page 691)
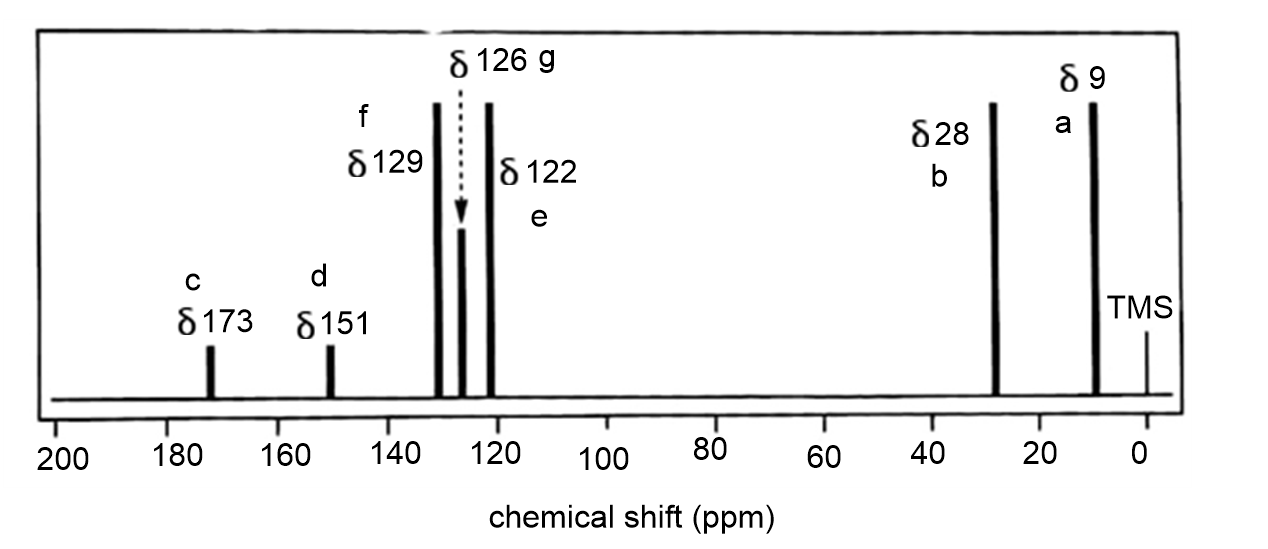
The standard 13CNMR spectrum of phenyl propanoate is shown here. Predict the appearance of the DEPT-90 and DEPT-135 spectra.
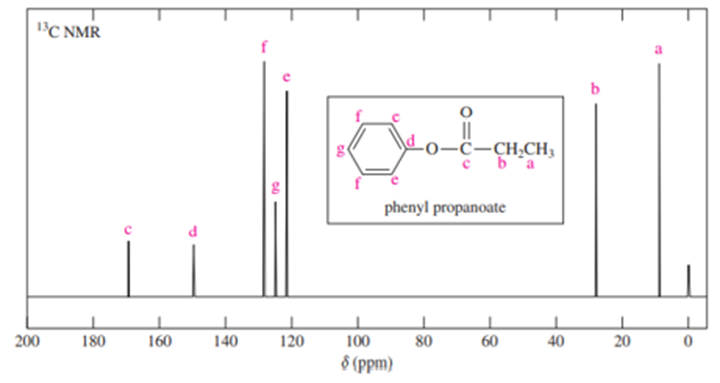
Short Answer
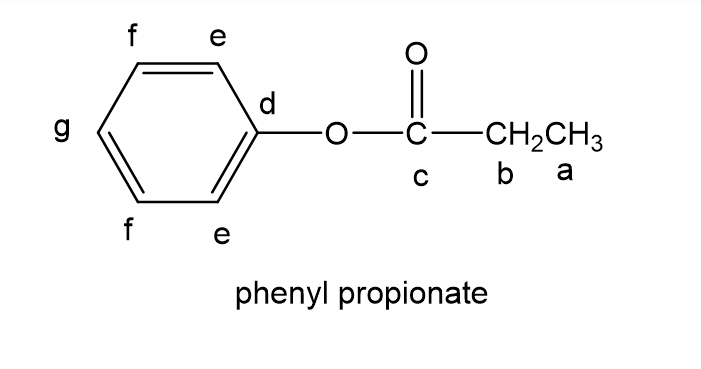
The full carbon spectrum of phenyl propanoate is presented below:
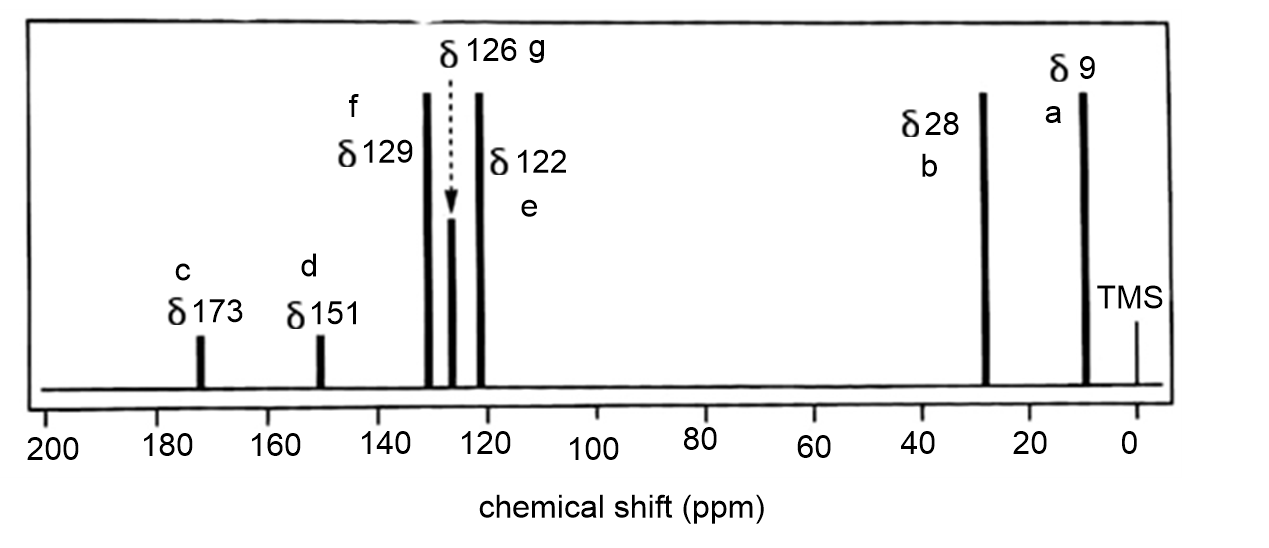
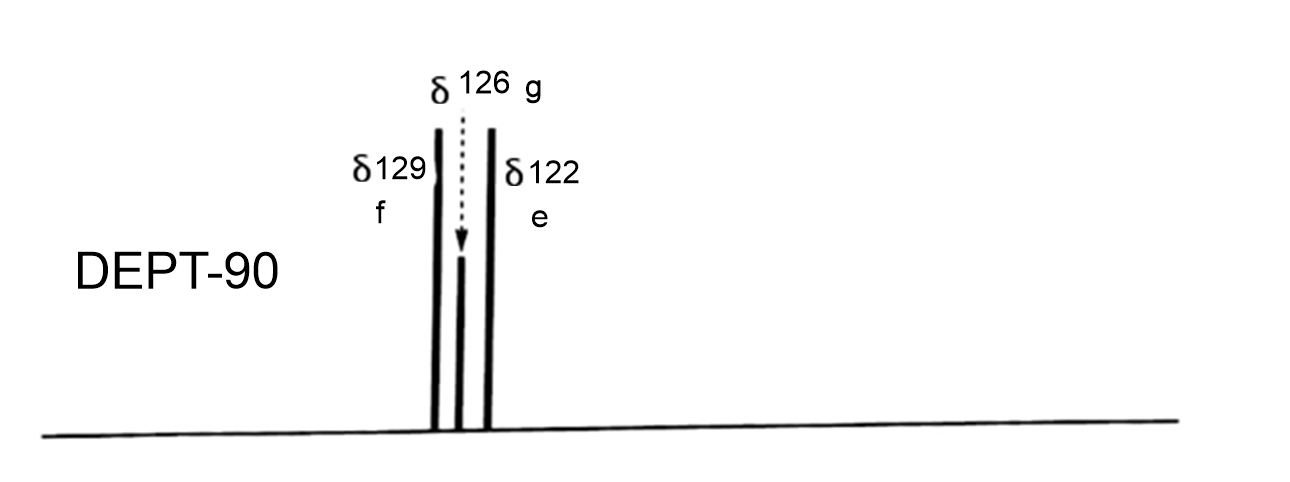
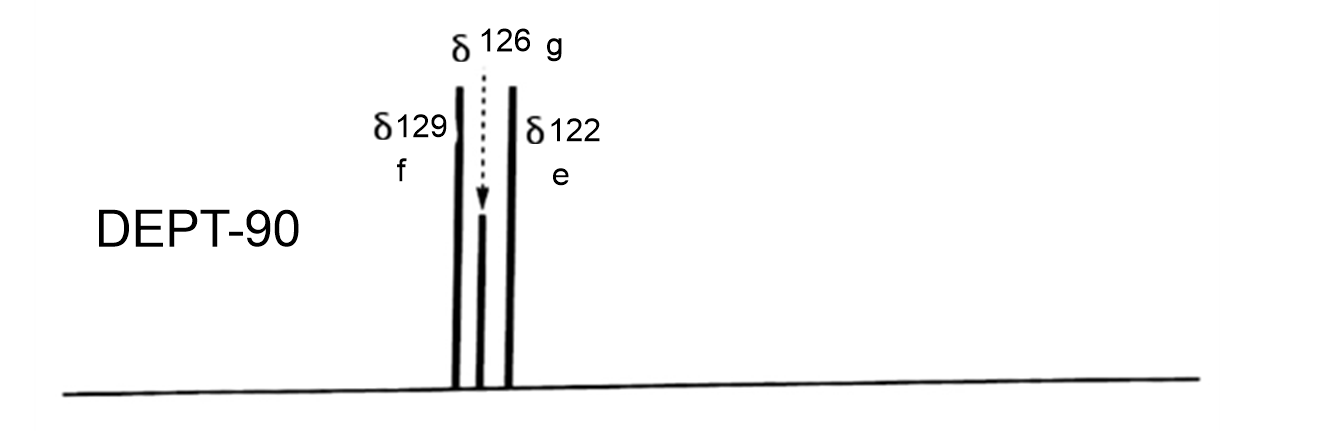
DEPT-90 will show only the methine carbons and all other peaks disappear.
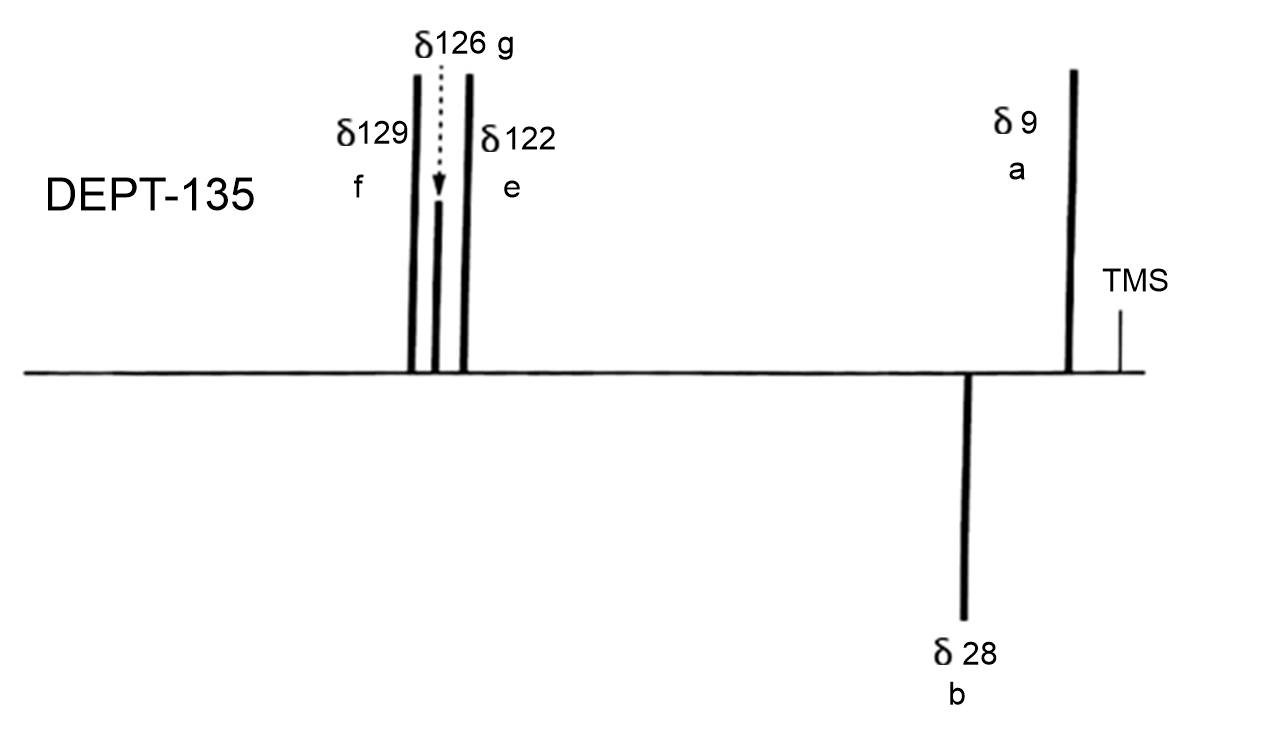
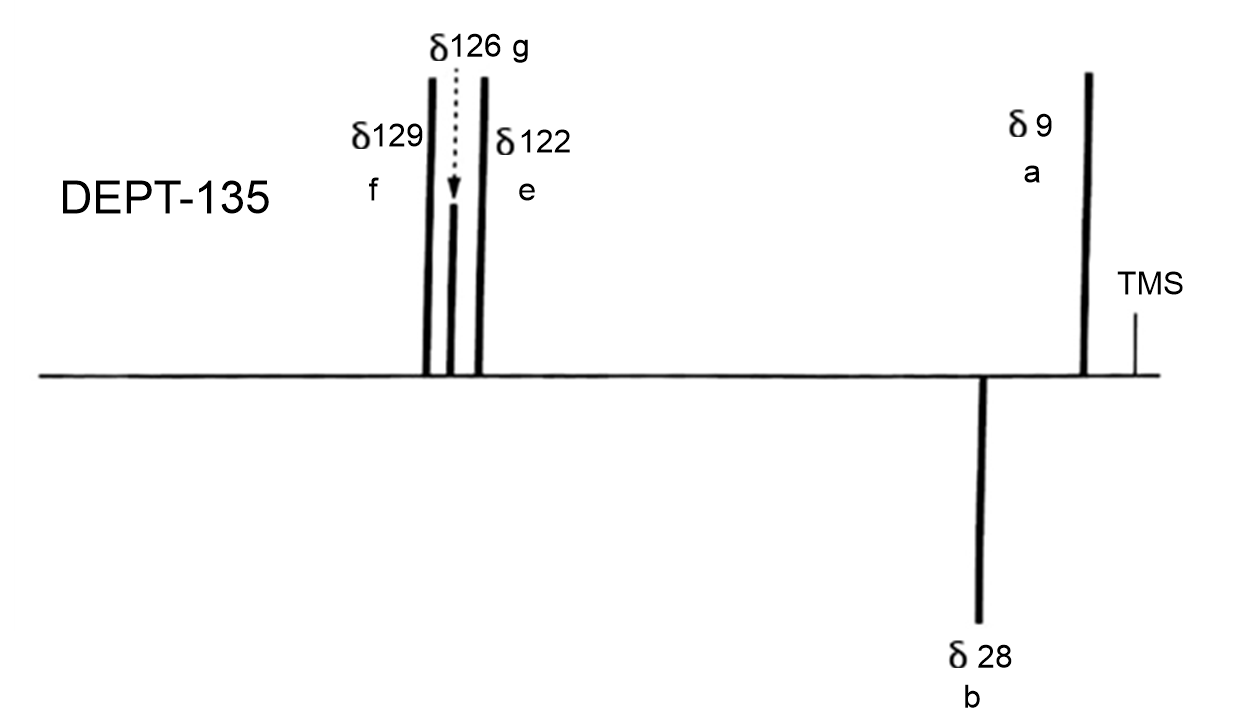
The DEPT-135 will show only the methyl and methine peaks pointed upwards and the methylene peaks pointed downwards.