Chapter 13: Q2P (page 656)
Predict the chemical shifts of the protons in the following compounds
Learning Materials
Features
Discover
Chapter 13: Q2P (page 656)
Predict the chemical shifts of the protons in the following compounds






All the tools & learning materials you need for study success - in one app.
Get started for free
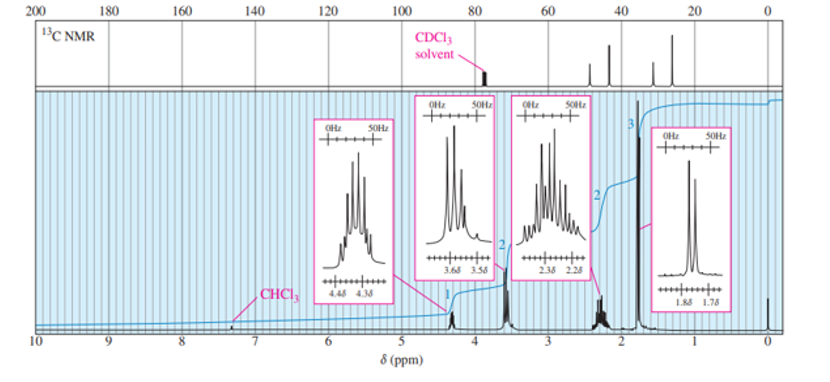
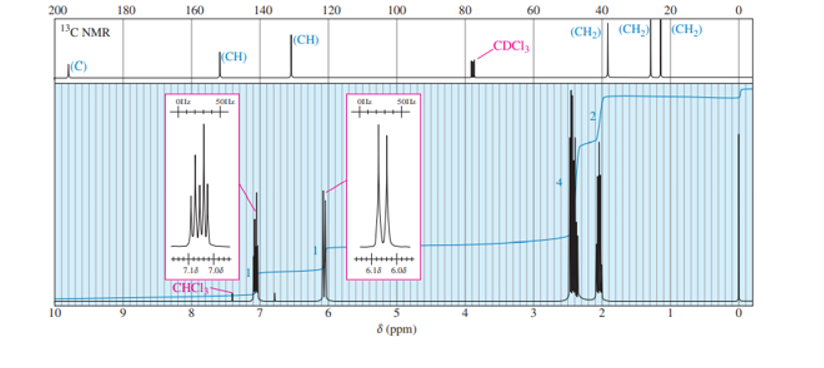
A small pilot was adding bromine across the double bond of but-2-ene to make 2,3-dibromobutane. A controller malfunction allowed the reaction temperature to rise beyond safe limits. A careful distillation of the product showed that several impurities had formed, including the one having the NMR spectra that appear below. Determine its structure and assign the peaks to the protons in your structure.
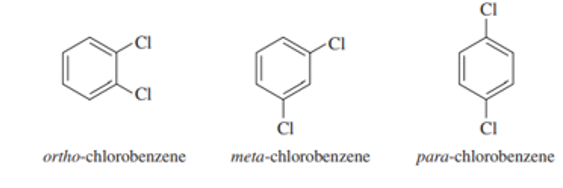
The three isomers of dichlorobenzene are commonly named ortho-chlorobenzene, meta-chlorobenzene, and para-chlorobenzene. These three isomers are difficult to distinguish using proton NMR, but they are instantly identifiable usingNMR.
Propose mechanisms to show the interchange of protons between ethanol molecules under
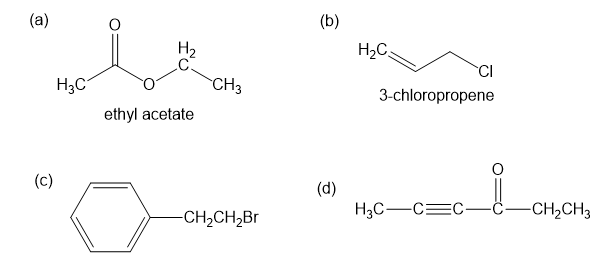
For each compound shown below,
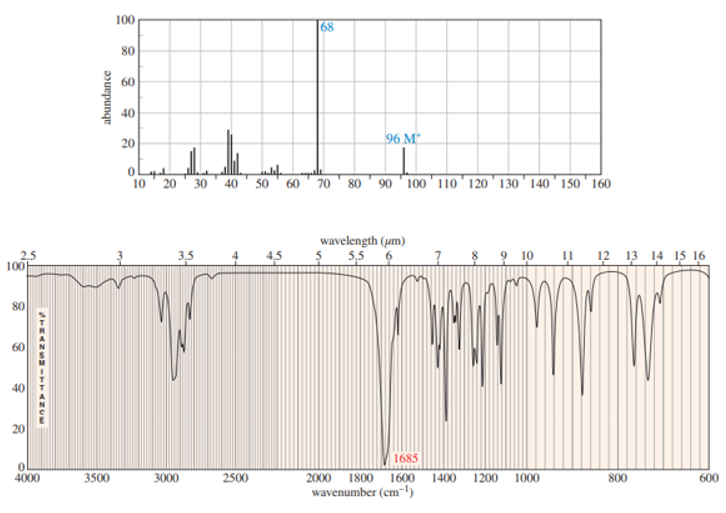
The following spectra are taken from a compound that is an important starting material for organic synthesis. Determine the structure, first by considering each spectrum individually, and then by considering all the spectra together. Assign peaks to show that your proposed structure accounts for all the major features of each spectrum. DEPT information is given in blue on the carbon NMR
What do you think about this solution?
We value your feedback to improve our textbook solutions.