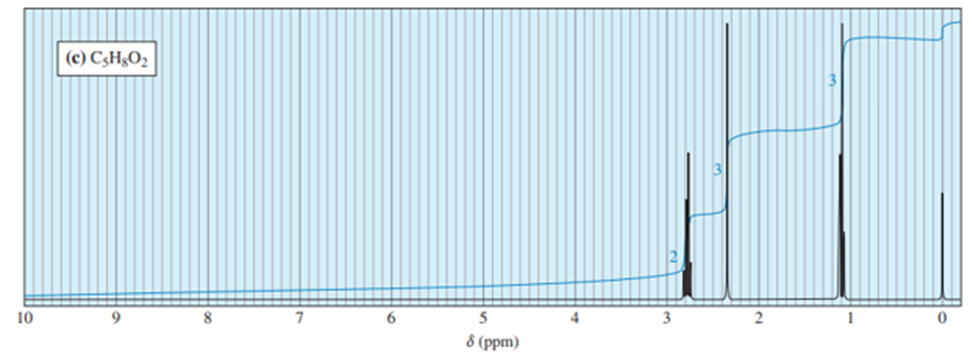
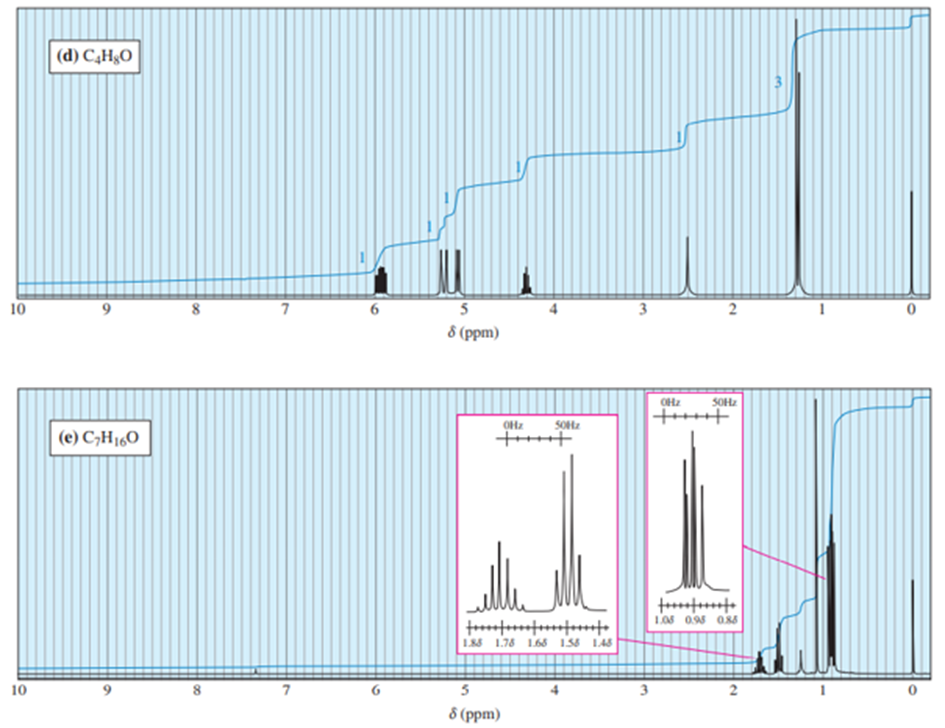
Chapter 13: Q41P (page 702)
For each compound shown below,
- Sketch theNMR spectrum (totally decoupled, with a singlet for each type of carbon), showing approximate chemical shifts.
- Show the multiplicity expected for each signal in the off-resonance-decoupled spectrum.
- Sketch the spectra expected using the DEPT-90 and DEPT-135 techniques.
Short Answer
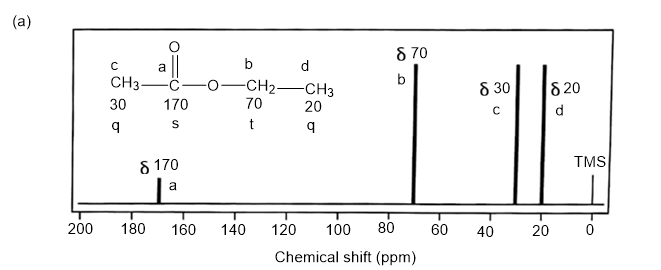
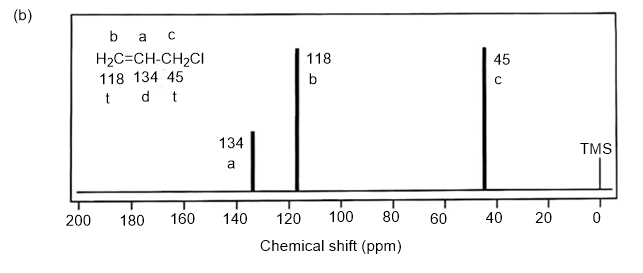
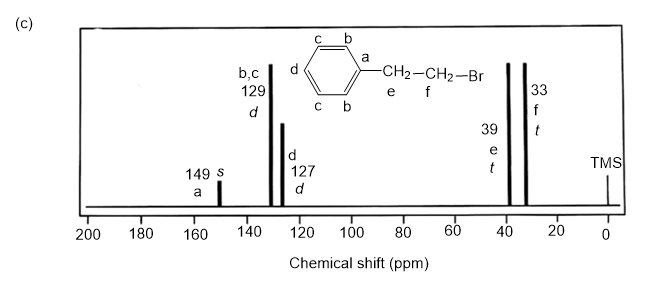
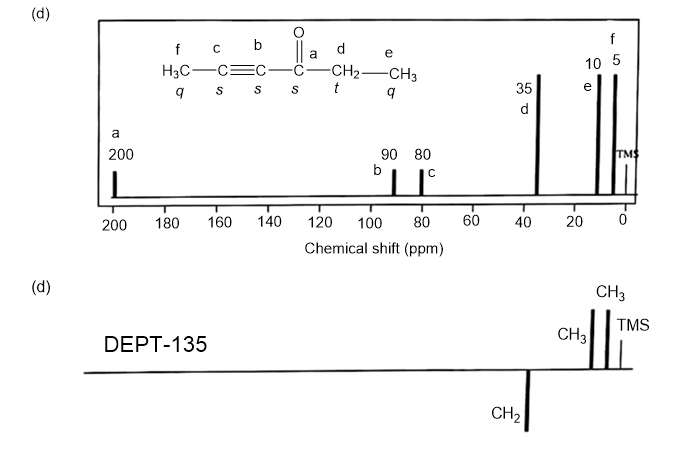
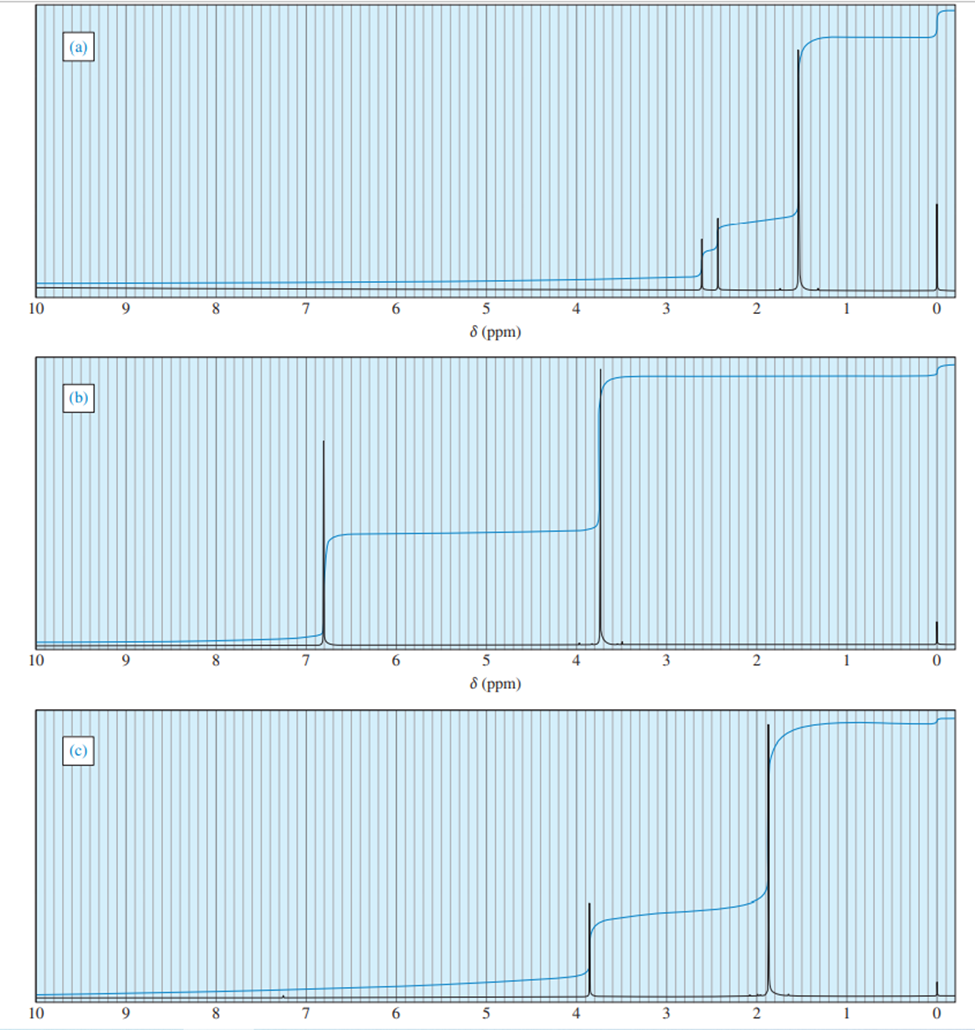
Answer. 1. and 2.
NMR spectrums for the given molecules have been sketched and multiplicities have been assigned along with chemical shift values in the spectrum. “s” means singlet, “d” means doublet, “t” means triplet and “q” means quartet.
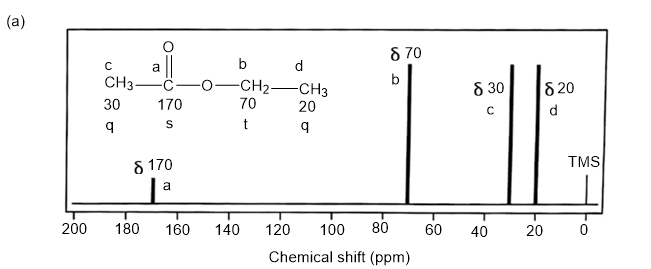
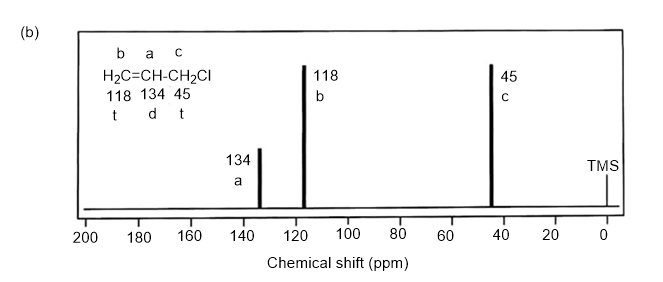
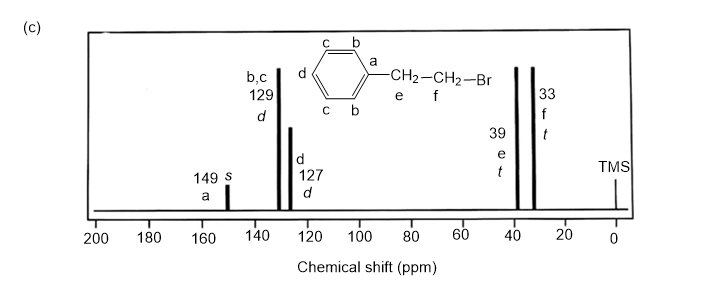
Splitting multiplicity has been written in italic alphabets so as to avoid confusion.
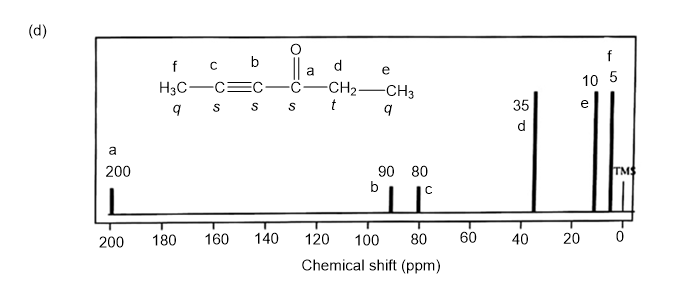
Spin-multiplicity has been written in italic alphabets so as to avoid confusion.
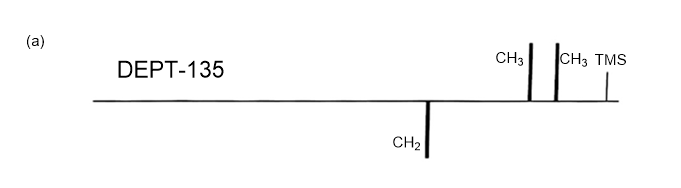
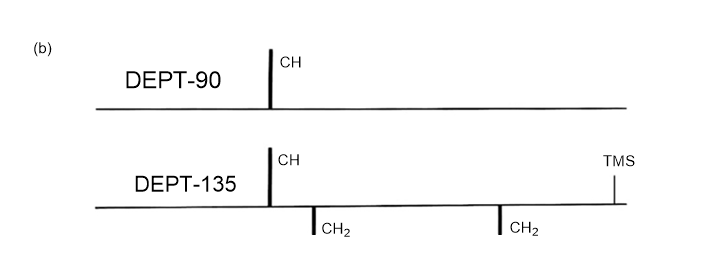
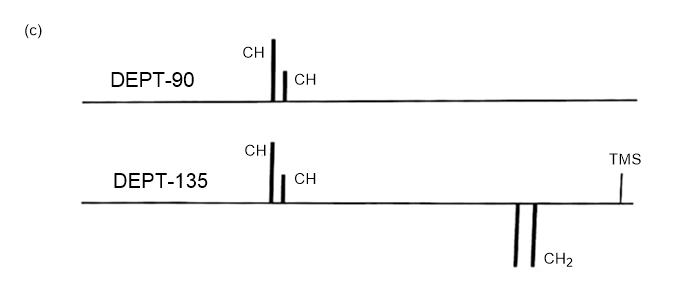
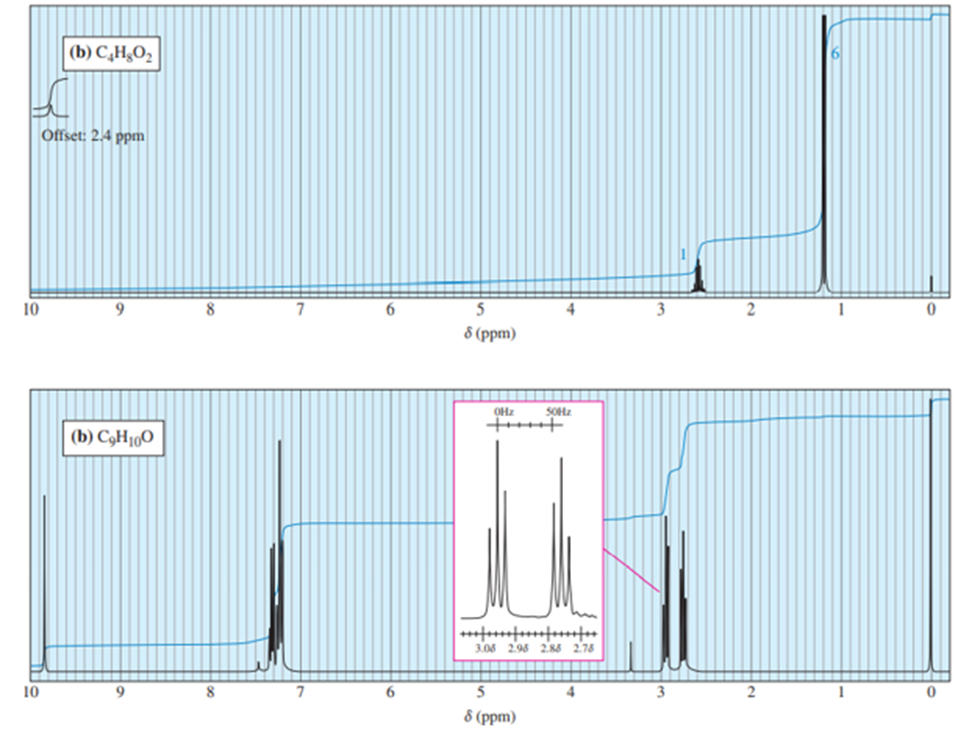
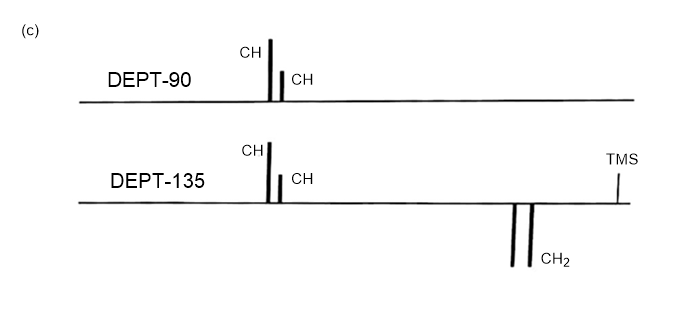
Answer 3.DEPT-90 and DEPT-135 spectra for the given molecules have been sketched.
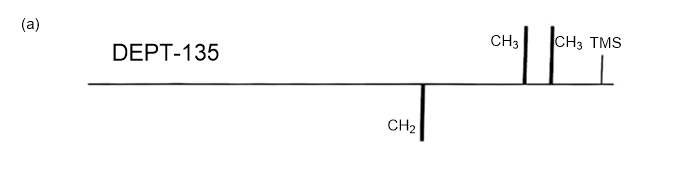
The DEPT-90 spectrum for ethyl acetate would have no peaks because there are no methine groups.
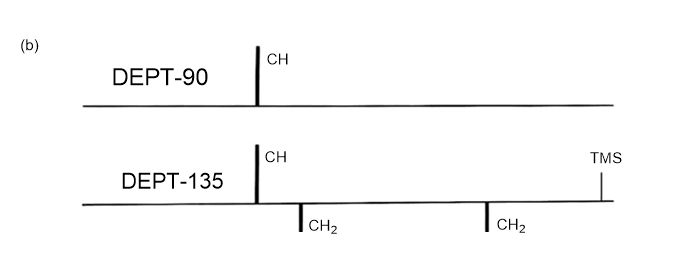

The DEPT-90 spectrum would have no peaks because there are no methine groups in molecule (d).