Chapter 13: Q40P (page 702)
Tell precisely how you would use the proton NMR spectra to distinguish between the following pairs of compounds.
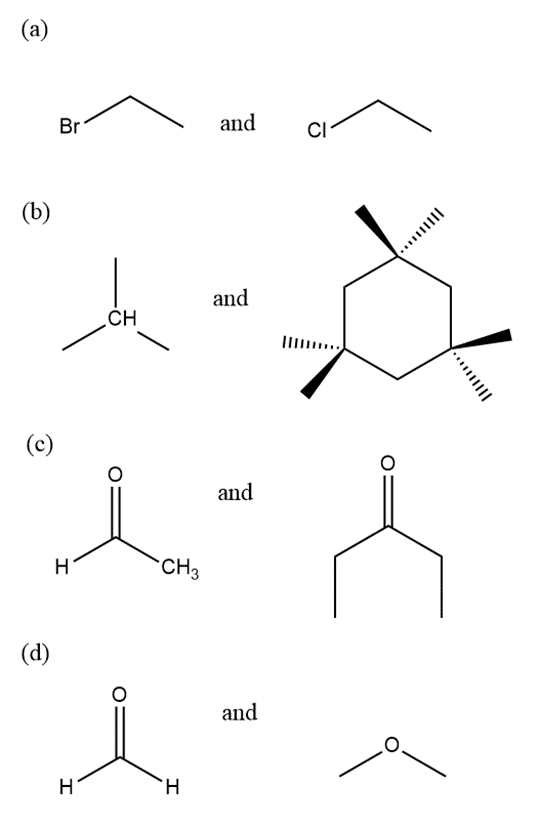
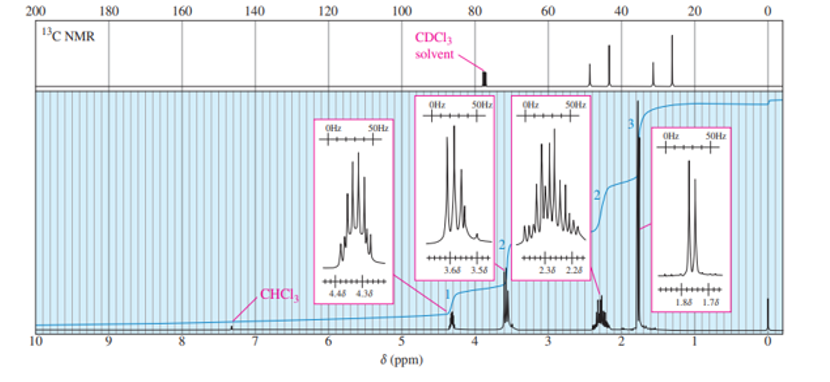
Short Answer
(a) Based on chemical shift values
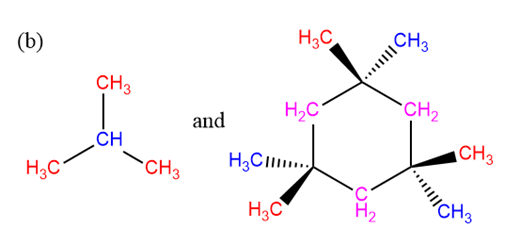
(b) Based on the number of signals
(c) Based on the chemical shift values
(d) Based on the chemical shift values



 and four dibrominated products
and four dibrominated products . These products are easily separated by GC-MS, but the dibrominated products are difficult to distinguish by their mass spectra. Draw the monobrominated product and the four dibrominated products and explain how
. These products are easily separated by GC-MS, but the dibrominated products are difficult to distinguish by their mass spectra. Draw the monobrominated product and the four dibrominated products and explain how NMR would easily distinguish among these compounds.
NMR would easily distinguish among these compounds.