Chapter 13: Q3P (page 658)
Determine the number of different kinds of protons in each compounds
- 1-bromopropane (b) 2-bromopropane
- 2,2-dimethylpropane (d) 2,2-dimethylpentane
- (e) 1-chloro-4-methylbenzene (f) 1-chloro-2-methylbenzene
Learning Materials
Features
Discover
Chapter 13: Q3P (page 658)
Determine the number of different kinds of protons in each compounds
All the tools & learning materials you need for study success - in one app.
Get started for free
The three isomers of dichlorobenzene are commonly named ortho-chlorobenzene, meta-chlorobenzene, and para-chlorobenzene. These three isomers are difficult to distinguish using proton NMR, but they are instantly identifiable using 13C NMR
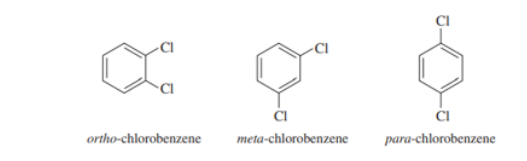
Each of these four structures has molecular formula . Match the structure with its characteristic proton NMR signals. (Not all of the signals are listed in each case.)
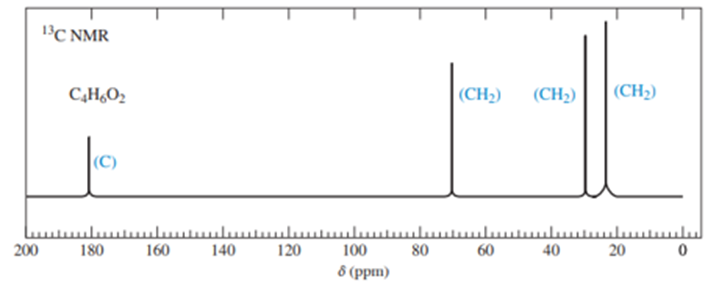
An inexperienced graduate student was making some 4-hydroxybutanoic acid. He obtained an excellent yield of a different compound, whose 13CNMR spectrum is shown here.
Hexamethylbenzene undergoes radical bromination with N-bromosuccinimide to give one monobrominated product and four dibrominated products
and four dibrominated products . These products are easily separated by GC-MS, but the dibrominated products are difficult to distinguish by their mass spectra. Draw the monobrominated product and the four dibrominated products and explain how
. These products are easily separated by GC-MS, but the dibrominated products are difficult to distinguish by their mass spectra. Draw the monobrominated product and the four dibrominated products and explain how NMR would easily distinguish among these compounds.
NMR would easily distinguish among these compounds.
Question:Using a 60-MHz spectrometer, a chemist observes the following absorption: doublet, J = 7 Hz, at 4.00
(a) What would the chemical shift () be in the 500-MHz spectrum?
(b) What would the splitting value J be in the 500-MHz spectrum?
(c) How many hertz from the TMS peak is this absorption in the 60-MHz spectrum? In the 500-MHz spectrum?
What do you think about this solution?
We value your feedback to improve our textbook solutions.