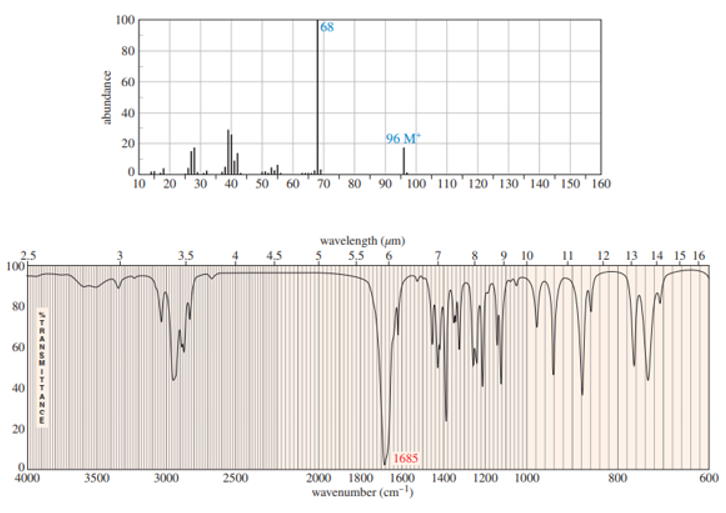
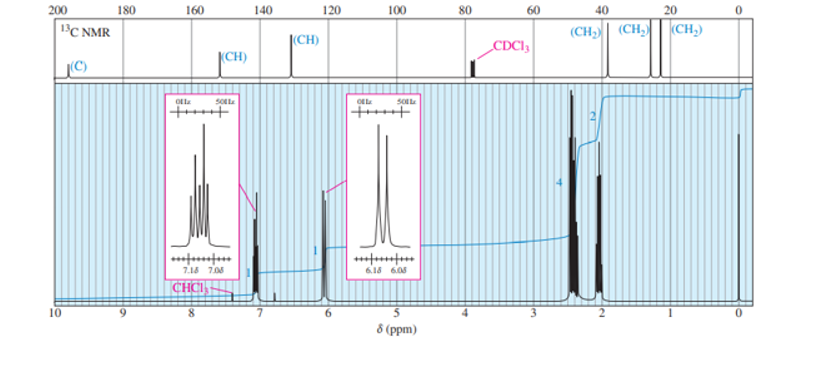
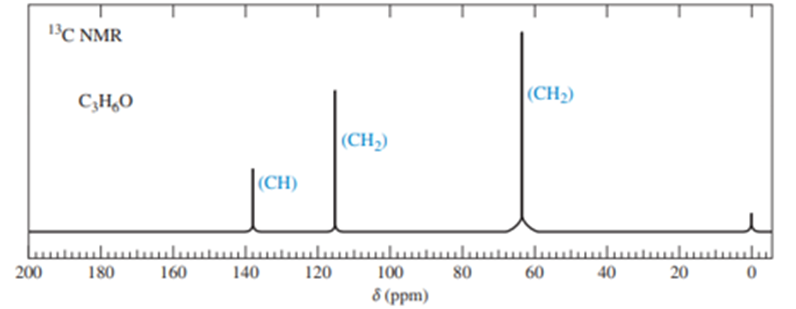
Chapter 13: Q37P (page 702)
Question:Using a 60-MHz spectrometer, a chemist observes the following absorption: doublet, J = 7 Hz, at 4.00
(a) What would the chemical shift () be in the 500-MHz spectrum?
(b) What would the splitting value J be in the 500-MHz spectrum?
(c) How many hertz from the TMS peak is this absorption in the 60-MHz spectrum? In the 500-MHz spectrum?
Short Answer
Answer
- The chemical shift of remains the same irrespective of the operating frequency of the spectrometer.
- The splitting value J= 7 Hz.
- For 60 MHz, the operating frequency is 240 Hz and for 500 MHz the operating frequency is 2000 Hz.