Chapter 13: Q.13-42P (page 703)
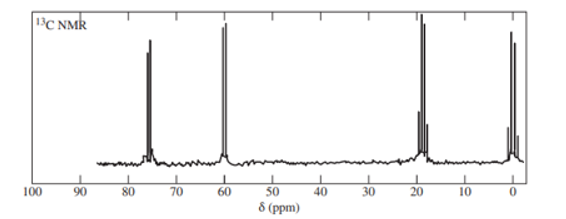
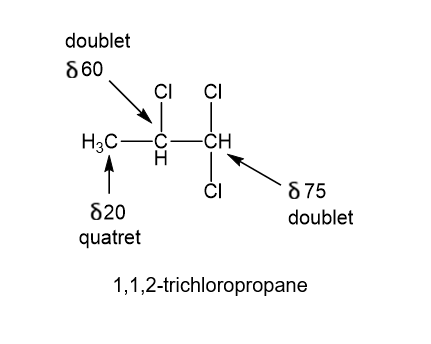
The following off-resonance-decoupled carbon NMR was obtained from a compound of formula. Propose a structure for this compound and show which carbon atoms give rise to which peaks in the spectrum.
Short Answer
Learning Materials
Features
Discover
Chapter 13: Q.13-42P (page 703)
The following off-resonance-decoupled carbon NMR was obtained from a compound of formula. Propose a structure for this compound and show which carbon atoms give rise to which peaks in the spectrum.


All the tools & learning materials you need for study success - in one app.
Get started for free
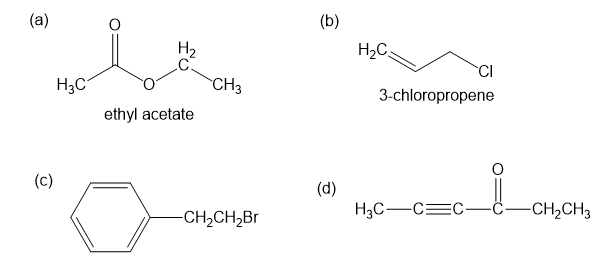
For each compound shown below,
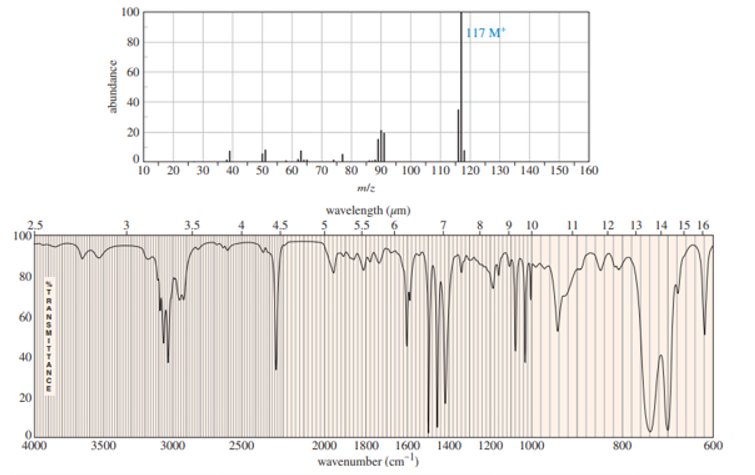
A compound was isolated as a minor constituent in an extract from garden cress. Its spectra are shown here.
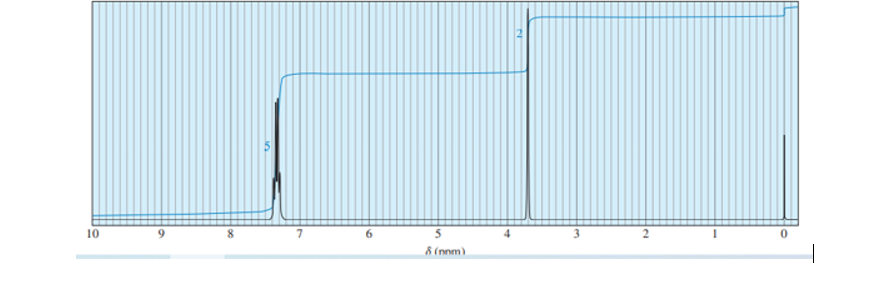
Question: Give the spectral assignments for the protons in isobutyl alcohol (Solved Problem 13-4). For example, Ha is a singlet, area = 1, at 2.4
The three isomers of dichlorobenzene are commonly named ortho-chlorobenzene, meta-chlorobenzene, and para-chlorobenzene. These three isomers are difficult to distinguish using proton NMR, but they are instantly identifiable usingNMR.
Question: An unknown compound has the molecular formula C9H11Br. Its proton NMR spectrum shows the following absorptions: singlet, d7.1, integral 44 mm singlet, d2.3, integral 130 mm singlet, d2.2, integral 67 mm Propose a structure for this compound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.